RG Dr. Daniel Schröder
Welcome to the website of the junior research group Dr. Daniel Schröder!
- Current notice
-
From April 1st, 2021 we are located at TU Braunschweig: https://www.tu-braunschweig.de/ines/
- Selected results
March 2020
The commercialization of pure metals as anode material is often called the "Holy Grail" in battery research. This statement is based on the fact that the energy density of batteries can be increased significantly by the use of pure metals. However, several challenges still need to be overcome before metal anodes can be commercialized. One of the main challenges is to prevent the growth of so-called dendrites during battery charging. Within the scope of several study projects, the WG Schröder works on the analysis and prevention of dendrites. In her bachelor thesis, Ronja Haas reduced the growth of dendrites by the use of different sodium metal alloys. For this purpose, she used liquid sodium-potassium alloys as an anode and sodium-tin alloys as protective layer. Julian Kreissl's master thesis also dealt with the prevention of dendrites in sodium-oxygen batteries. In cooperation with the group of Prof. Dr. Peter R. Schreiner, he used functionalized diamondoids as an additive in the electrolyte. These "molecular diamonds" are incorporated into the anode during charging and ensure planar metal deposition (Figure a; 10.1002/cssc.201903499). Both theses were awarded first place in the categories "Best Bachelor Thesis" and "Best Master Thesis" as part of the Students Program for Electric Mobility of the German Federal Ministry of Education and Research (BMBF) and the Fraunhofer-Gesellschaft – DRIVE-E (Figure b; https://www.uni-giessen.de/ueber-uns/pressestelle/pm/pm188-19driveepreisfuerwissenschaftlichennachwuchs; https://www.iisb.fraunhofer.de/en/press_media/press_releases/pressearchiv/archiv_2019/drive-e_2019_studienpreise.html).
July 2020
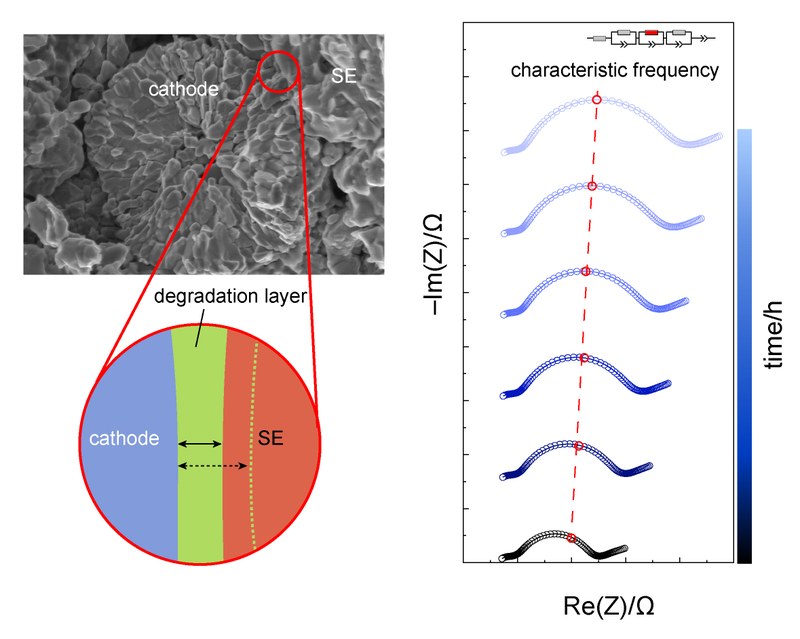
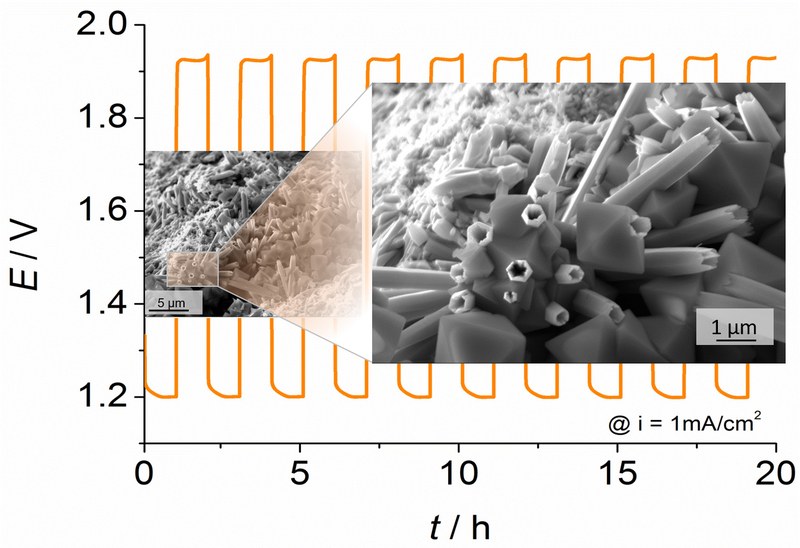
Lithium-ion batteries have received considerable attention due to the increasing demand for electrochemical energy storage. However, commercial lithium-ion batteries suffer from severe safety concerns stemming from the organic liquid electrolyte (e.g., they are flammable). Solid-state batteries (SSBs) are expected to be a promising candidate complementing batteries with organic liquid electrolyte. Nevertheless, the development of SSBs still faces considerable challenges hindering their full-scale commercialization. One of the major issues is the high charge-transfer resistance between the electrode materials and the solid electrolyte due side reactions at the electrode/electrolyte interface. Therefore, the WG Schröder and the WG Janek investigate the underlying degradation processes in SSBs. In detail, we examine the degradation reactions at the interface of various composite cathode materials and ceramic solid electrolytes within the GER-JPN-joint BMBF-project InCa. The SEM image displays the interface between composite cathode and solid electrolyte, and the graphic schematically illustrates the degradation process. Impedance measurements were carried out to monitor the evolution of the degradation layer between the compounds. A characteristic frequency (~200 – 500 Hz), which can be ascribed to the degradation processes at the interface, is indicated in the Nyquist plot. Thereby, the gradually increasing resistance between composite cathode and solid electrolyte are mainly caused by the growing degradation layer. (Picture submitted by TongTong Zuo and Daniel Schröder).
October 2020
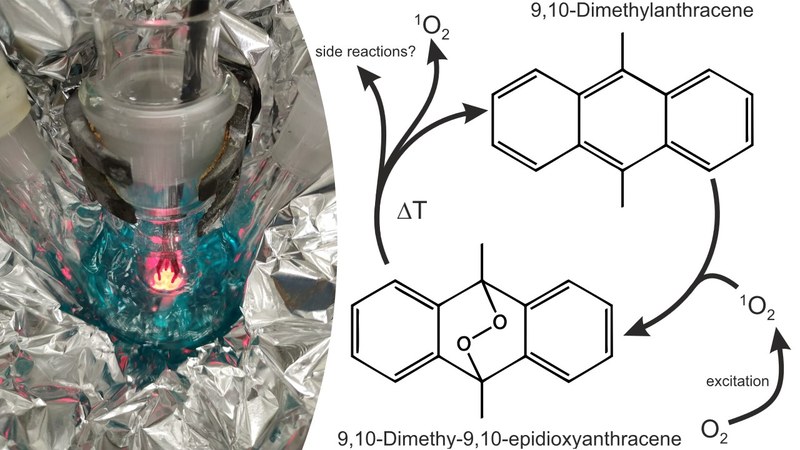
Due to their high theoretical storage capacity, lithium-oxygen batteries are regarded as possible energy storage systems of the future, both for mobile and stationary applications. In them, metallic lithium (anodic reaction) is converted with oxygen (cathode reaction) to lithium peroxide. The lithium peroxide formed is stored in the battery and converted back into oxygen and metallic lithium when charged. Despite intensive research in recent years, this type of battery is not yet able to achieve high cycle numbers for electrical recharging. This is due to various side reactions during the charging and discharging process, the origin of which has not yet been fully understood. A possible source of the unintentionally generated by-products is singlet oxygen (¹O₂) formed during operation. ¹O₂ is an excited, short-lived variant of molecular oxygen. It is a strong oxidant and reacts with the other components of the battery such as the electrolyte and the electrode materials. However, since ¹O₂ is not long-term stable and reacts after only a few milliseconds, detection is difficult. One possibility for detection is the so-called trapping in which ¹O₂ is reacted with other molecules, which can then be detected later. At the AG Schröder, the molecule 9,10-dimethylanthracene (DMA) shown in the figure is currently being investigated as an agent for detecting 1O2 in lithium-oxygen batteries, since it reacts specifically with singlet oxygen to form 9,10-dimethyl-9,10-epidioxyanthracene (DMA-O₂). Little is known about its stability and its influence on the chemistry of lithium-oxygen batteries. The DMA-O₂ required for this is produced photochemically in the AG Schröder (see picture) and then purified before it is further investigated. (Image by: Adrian Schürmann)
December 2020
In recent years, organic redox flow batteries (RFB) have increasingly become the focus of attention as large-scale energy storage systems, since they do not contain toxic and rare metal ions compared to the established vanadium RFB. Due to this, the demand for new organic active materials, which are used as catholyte or anolyte in organic RFB, is increasing. The long-term stability of the organic active materials is crucial for a possible commercial success of RFB. Our research group is focusing on the characterization of the performance and stability of organic active materials for RFB in operation. The picture shows an example of the results obtained with a diazanaphthoquinone (synthesized by AG Wegner at the JLU; cooperation within a project funded by the BMEL). The diazanaphthoquinone (top) was measured using an operando UV/Vis cell (left), while electrochemical measurements (right) were performed simultaneously. The UV/Vis measurements are used to identify products and degradation products, whereas long-term electrochemical measurements allow to determine the kinetics of the occurring degradation reactions. Overall, our group always relies on a powerful combination of analytics (operando, in-situ and ex-situ) and theoretical considerations to advance the understanding of organic active materials for RFB. (Image by: Dominik Emmel and Simon Kunz)
April 2016
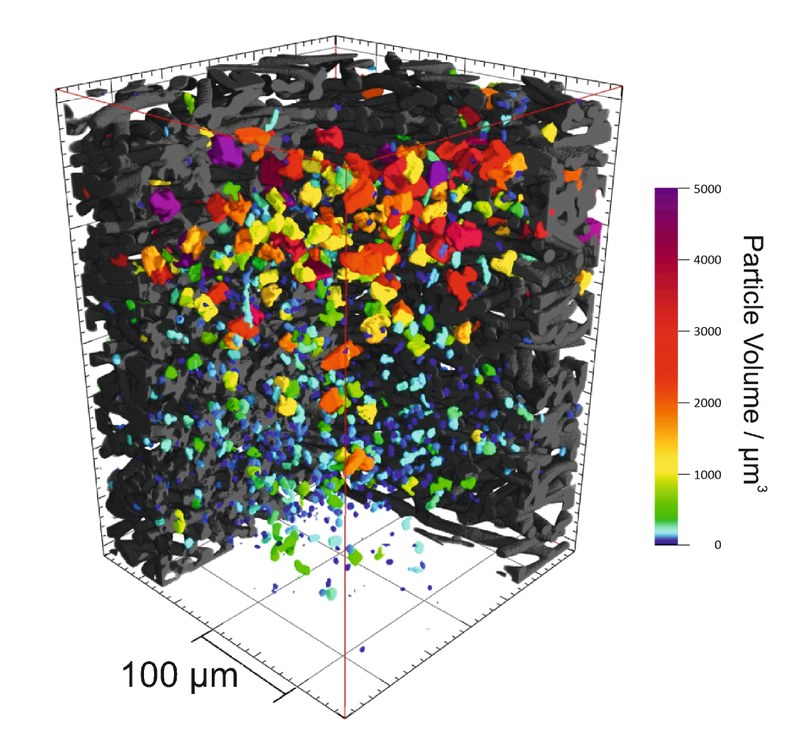
Peeking inside batteries with X-rays – We show the cathode of a Na/O2 battery in which sodium superoxide is deposited as discharge product. Sodium superoxide is usually observed as cubical particles with up to 20 µm particle size, but can not be monitored during operation directly inside the battery because it is covered by the battery housing and other materials. We visualize these particles (color gradient as measure for particle volume) by means of synchrotron X-ray transmission tomography of a fully discharge battery cathode. Measurements were conducted in cooperation with the Helmholtz-Zentrum Berlin für Materialien und Energie at the electron storage ring BESSY II, and were recently published (http://www.nature.com/articles/srep24288).Electrically rechargeable metal/oxygen batteries, such as the Na/O2 battery, have been considered as possible future energy storage devices. Metal/oxygen batteries are investigated intensively in the Janek research group within the framework of several third-party funded research projects, such as the BASF-Network “Electrochemistry and Batteries”, and the BMBF-Project “BenchBatt”. Main focus is thereby on gaining understanding of the electrochemical and chemical processes and thus on improving cell performance. (Picture submitted by Conrad Bender and Daniel Schröder.)
October 2016
In view of the fact that the establishment of alternative energy sources is coupled to the storage of the so generated energy different concepts of stationary energy storage are being investigated at the moment. Besides conventional battery systems like lithium-ion batteries alternative storage solutions that are based on abundant and cost efficient materials enter the limelight. For example several organic molecules that can be obtained by many kinds of resources can be optimized for their application in electrochemical cells. The desired molecules for this application are being defined within the scope of a collaboration between the Physical-Chemical- and the Organic Institute of the Justus-Liebig University by the investigation of the correlation between their structure and electrochemical properties. One important way to have an influence on these properties is to vary the pH value of the electrolyte the electrochemically active species is dissolved in. By modifying it the potential of the electron transfer can be varied to optimize the compound for the application in different environments. The image shows the influence of the pH value on the compounds charge transfer characteristic by shifting its potential. This is illustrated by cyclic voltammogramms that have been measured versus a Ag/AgCl reference electrode within a three-electrode assembly. (Picture submitted by Jonas Hofmann.)
October 2017
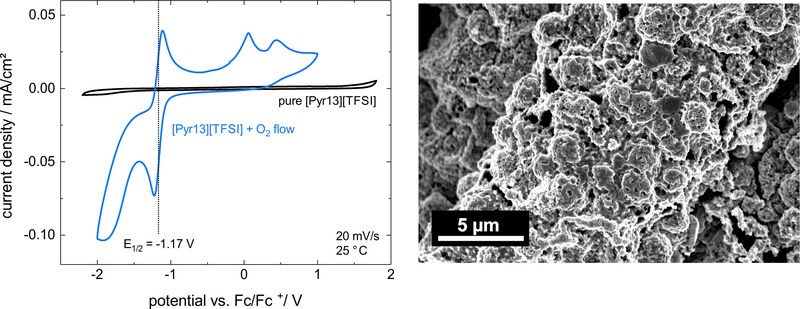
The electrochemical synthesis from Ionic Liquids (ILs) offers an alternative route for the formation of unusual noble metal compounds, like silver oxides. A major advantage of this route is the possibility to work in the absence of harsh conditions like high pressures or temperatures and the use of highly reactive and/or hazardous educts. This approach is analogous to the reactions in a metal air battery, where oxygen is reduced to superoxide at the cathode (Red1: O2 + e– → O2–) and metal oxidized to the corresponding metal ions (Ox, here: Ag → Ag+ + e–). These can precipitate together as a metal oxide (here: AgxOy) and/or undergo further disproportionation. However, the metal ions compete with the oxygen of the reduction at the cathode and the inverse reaction to Ox can occur (Red2: Ag+ + e– → Ag) instead of the wanted oxygen reduction reaction (ORR).A pure oxygen saturated IL ([Pyr13][TFSI]) shows a clear ORR signal (Red1) in the cyclic voltammogram at -1.17 V (left figure, blue line). After the addition of the corresponding silver salt (Ag[TFSI]) the peak vanishes and another redox potential at -0.38 V appears, which corresponds to the Ag/Ag+ potential. Thus, unlike in metal air cells, no reaction of oxygen and silver to the metal oxide (Red1 and Ox) is observable. Instead, pure silver is deposited on the cathode and the ORR is suppressed (Red2 and Ox). The right figure shows the porous morphology of the deposited silver.The major target of this project is a better understanding of the occurring reduction reactions Red1 and Red2 depending on parameters like temperature, scan rate, potential, O2- and Ag-concentration. (Picture submitted by Peter Schmitz)
February 2018
Do we always get ZnO as discharge product in electrically rechargeable zinc–air batteries? Nowadays, air breathing batteries appear as a good alternative to overcome our increasing demand of energy. One of the best candidates regarding its cost of production, recyclability and safety is the electrically rechargeable Zn/air battery. Therefore, the development of the rechargeable Zn/air battery has attracted much attention and received an intense research effort to extend its cycling stability. One key issue in alkaline electrically rechargeable Zn/air battery is the electrochemical reaction at the Zn electrode. ZnO as discharge product cannot be fully converted to Zn. In general, the formation of ZnO is influenced by many parameters such as local concentration, pH and diffusion of Zn ions species in alkaline solution. However, the nucleation and growth of ZnO in electrically rechargeable Zn/air battery can be also influenced by an interaction between discharge and charge process. We observed that a battery component, such as a Sn current collector, could also affect the nature of the discharge product during cycling. The scanning electron micrograph (figure below) shows orthorhombic ZnSnO3 or Zn2SnO4 crystallites surrounded by many hexagonal tubes of ZnO obtained after 25 cycles on top of the Zn electrode. These new investigations are part of the ongoing German-Japanese BMBF joint project ‘Zisabi’ to gain a deeper insight into electrically rechargeable Zn/air batteries. (Picture submitted by Saustin Dongmo and Daniel Schroeder.)
November 2018
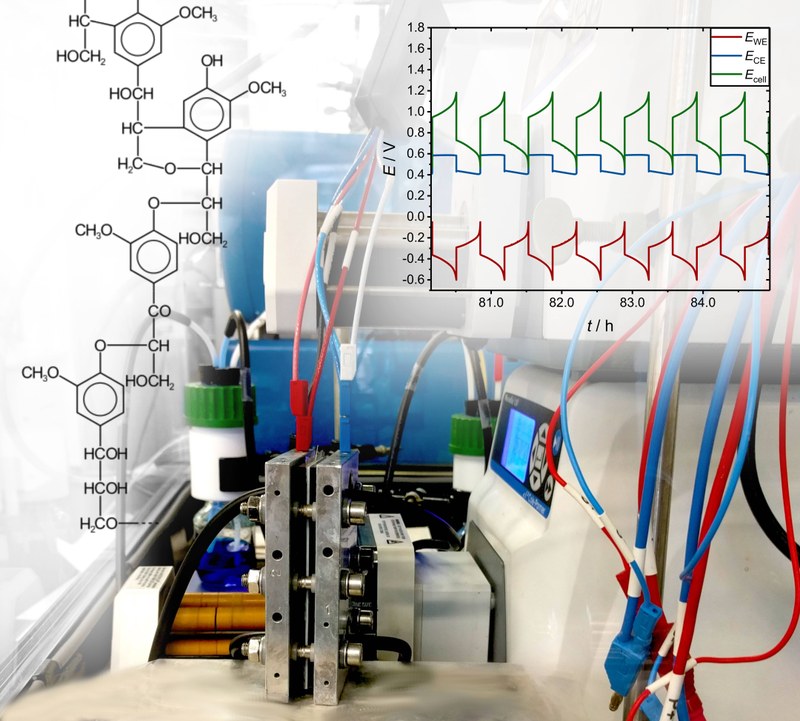
Redox flow batteries are suitable for the stationary storage of intermittent energy provided by renewable energies. The beneficial design of the redox flow battery enables the storage of energy in liquid electrolytes in different oxidation states outside the actual electrochemical cell. Currently, the active material dissolved in the electrolyte constitutes the main cost factor for redox flow batteries. Using cheaper organic molecules instead, that for example may be obtained by purification of waste products from the pulp and paper industry, could save costs and further increase the economic viability of redox flow batteries. In the search for a suitable molecule that fits this application, the BMEL project FOREST is currently investigating several organic molecules for their stability and performance. The picture shows the measurement setup used for a lab-sized redox flow battery (center). The dissolved active materials – here in the form of an organic electrolyte on the anode and a vanadium (IV) based electrolyte on the cathode side – are transported into the electrochemical cell by pumps. A reference electrode allows for the recording of the working (WE) and counter electrode (CE) potentials during battery cycling and therefore enables to draw conclusions about the performance and degradation behavior of the associated active materials (right). Picture submitted by Dominik Emmel and Jonas Hofmann, RG Dr. Daniel Schröder
July 2019
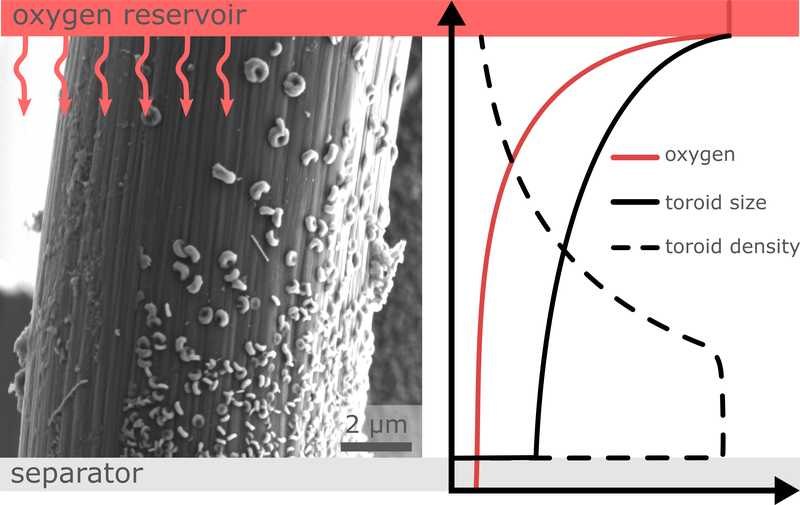
Rechargeable metal-oxygen batteries are considered as a potential technology in future energy storage systems. Alkali metal-oxygen batteries, such as lithium-oxygen batteries, are in particular focus of industry and research due to their high theoretical energy density. The lithium peroxide formed during discharge precipitates as a solid on the cathode structure, so the cathode design and understanding of the growth mechanism play a crucial role in achieving maximum energy densities. For this reason, the BMBF-project MeLuBatt – in close cooperation with the Institute for Energy and Systems Engineering (InES) at TU Braunschweig and the Fraunhofer Institute for Manufacturing Technology and Applied Materials Research (IFAM) in Oldenburg – is currently investigating the growth behaviour of the discharge product in the cathode of lithium oxygen batteries. The SEM image shows the magnification of a carbon fiber on which toroid-like Li2O2 particles have been formed. The size and particle density of these toroids strongly depends on the availability of dissolved oxygen in the electrolyte: With more dissolved oxygen (cathode side facing the O2 reservoir), fewer but significantly larger toroids are formed, while deeper in the electrolyte (cathode facing the separator side) the toroids are smaller, but grow in a higher density. The diagram schematically illustrates how the size and density of the toroids depend on the dissolved O2 in the electrolyte along a carbon fiber. (Picture submitted by Julian Kreißl, Daniel Langsdorf and Daniel Schröder).