Wegner Group
- AG Wegner
-
The understanding of structure-property relations is the key to create function on the molecular level. We combine concepts and tools of physical organic chemistry to address this challenge.
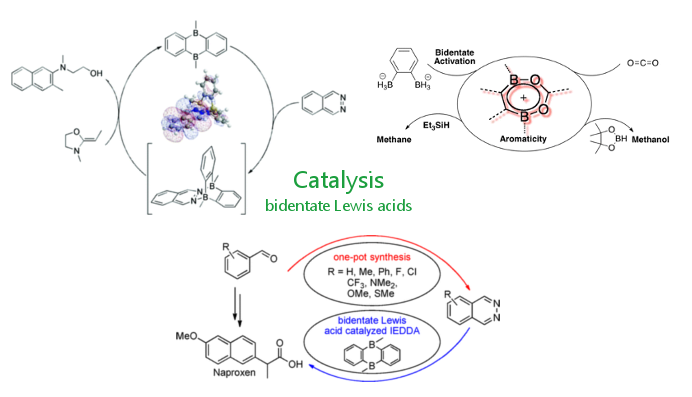
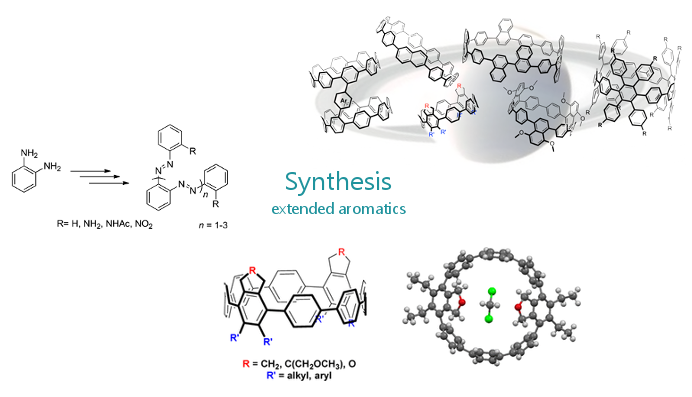
In this context we explore the unique opportunities to substitute carbon-carbon bounds with boron-nitrogen moieties in catalysis as well as new π-materials. Alternatively, we investigate new concepts to control properties of π-nanocarbons such as cycloparaphenylenes (CPPs) via synthesis in solution, via flow chemistry, as well as by on-surface-synthesis.
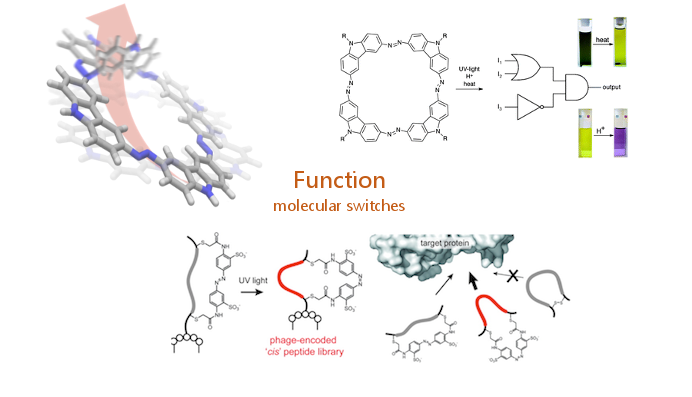
Additionally, we use dynamic strategies, namely molecular switches based on the azobenzene scaffold as molecular tool to address fundamental questions. These include London dispersion interactions, smart supramolecular assemblies for instance in macrocyclic arrangements.
In general we place our fundamental research in the context of energy-related questions, such as organic flow batteries, molecular solar thermal storage systems (MOST), metal-free hydrogen storage, or novel electronic organic materials.