Organocatalysis
Organocatalysis combines the concepts of molecular recognition as well as supramolecular chemistry with enzyme-like catalytic activity. Noting that about half of all enzymes do not carry a metal center it is obvious that this approach has long been underrated. Although this is a new and emerging field, it is already possible to catalyze many types of organic reactions with small, well-designed organic molecules. This circumvents the use of often toxic metals (leading to environmentally benign methods, green chemistry), and the preparation of the catalysts is much easier as it relies on the well-developed synthetic arsenal for tailor-making organic structures. In our group we have developed thiourea-based catalysts that are effective in catalyzing a variety of transformations such as Diels-Alder reactions, acetalization, THP-protection, stereoselective acyl transfer, reductions, epoxide openings and many more. Currently, we develop reactions and reaction sequences utilizing cooperative catalysis and multicatalysis.
- Director: Univ.-Prof. Peter R. Schreiner
- Organocatalysis
-
Current work:
Structure elucidation of aroma-active bicyclic benzofuran derivatives formed by Cystostereum murrayi. Fabio F. Brescia, Maximilian Koch, Raffael C. Wende, Peter R. Schreiner, Holger Zorn, Marco A. Fraatz
J. Agric. Food Chem. 2023, 71, 7744–7751. DOI: 10.1021/acs.jafc.3c01807.Among the monoterpenoid aroma compounds formed by the basidiomycete Cystostereum murrayi are highly potent bicyclic benzofuran derivatives. In addition to the dill ethers previously described in a few fungi, two stereoisomers of the rare 3,6-dimethyl-3a,4,5,6,7,7a-hexahydro-3H-1-benzofuran-2-one (1a and 2c), also known as dihydromenthofurolactones, and a C3-unsaturated analogue (3a) are formed by C. murrayi. The analysis of synthesized reference standards of the lactones allowed an unambiguous assignment of the stereoisomers formed by the fungus. Despite a similar structure, two key differences in the stereochemistry of the lactones and dill ethers emerged. The analysis of submerged cultures further revealed the formation of additional, so far unknown, fungal terpenoids, including limonen-10-ol (7) and the corresponding aldehyde limonen-10-al (8). Analysis of chiral terpenoids as well as supplementation studies, including stable isotope-labeled compounds, indicated independent biogenesis pathways for dill ethers and lactones.

Visible-Light-Induced Phenoxyl Radical based Metal–Organic Framework for Selective Photooxidation of Sulfides. Zhenghua Zhao, Kai Zhou, Minjie Liu, Lidong Guo, Yajing Shen, Dan Luo, Xin Hong, Zongbi Bao, Qiwei Yang, Qilong Ren, Peter R. Schreiner and Zhiguo Zhang
ACS Appl. Mater. Interfaces 2023, 15, 6982–6989. DOI: 10.1021/acsami.2c21304.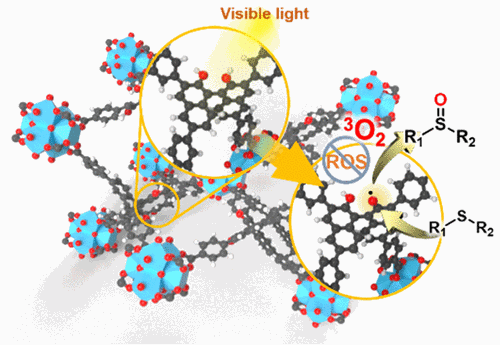
Phenoxyl radicals originating from phenols through oxidation or photoinduction are relatively stable and exhibit mild oxidative activity, which endows them with the potential for photocatalysis. Herein, a stable and recyclable metal–organic framework Zr-MOF-OH constructed of a binaphthol derivative ligand has been synthesized and functions as an efficient heterogeneous photocatalyst. Zr-MOF-OH shows fairly good catalytic activity and substrate compatibility toward the selective oxidation of sulfides to sulfoxides under visible light irradiation. Such irradiation of Zr-MOF-OH converts the phenolic hydroxyl groups of the binaphthol derivative ligand to phenoxyl radicals through excited state intramolecular proton transfer, and the excited state photocatalyst triggers the single-electron oxidation of the sulfide. No reactive oxygen species are produced in the photocatalytic process, and triplet O2 directly participates in the reaction, endowing Zr-MOF-OH with wide substrate compatibility and high selectivity, which also proposes a promising pathway for the direct activation of substrates via phenoxyl radicals.
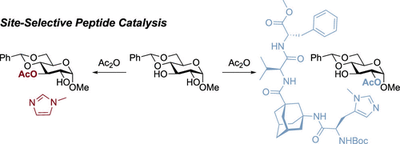
Site-Selective Acylation of Pyranosides with Immobilized Oligopeptide Catalysts. Alexander Seitz, Raffael C. Wende, and Peter R. Schreiner
Eur. J. 2023, 29, e202203002. DOI: 10.1002/chem.202203002.We report the site-selective acetylation of partially protected monosaccharides using immobilized oligopeptide catalysts, which are readily accessible via solid-phase peptide synthesis. The catalysts are able to invert the intrinsic selectivity, which was determined using N-methylimidazole, for a variety of pyranosides. We demonstrate that the catalysts are stable for multiple reaction cycles and can be easily reused after separation from the reaction solution. The catalysts can also be used in flow without loss of reactivity and selectivity.

Total Synthesis of (–)-Antroalbocin A Enabled by a Strain Release-Controlled Photochemical 1,3-Acyl Shift. Björn Siekmeyer, Dennis Lübken, Kevin Bajerke, Bastian Bernhardt, Peter R. Schreiner, and Markus Kalesse* Lett. 2022, 24, 5812–5816. DOI: 10.1021/acs.orglett.2c02347.
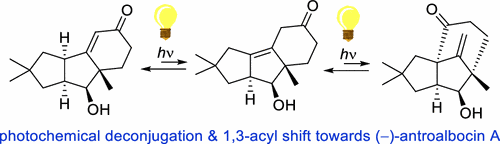
The first bioinspired, enantioselective, and protecting group free total synthesis of the antibacterial sesquiterpenoid (−)-antroalbocin A (1) has been achieved in 12 steps (5.4% overall yield) from dimedone. An organocatalytic Robinson annulation gave rapid access to the tricyclic enone (19) as starting material for the photochemical domino process of deconjugation and sigmatropic 1,3-acyl shift. Computational data of this process indicate that the 1,3-acyl shift benefits from the highly strained 1,3-enone 8. The transformation of 8 to its bridged isomer 5 is exergonic and, therefore, enables an increased conversion compared to unstrained substrates.
Synthetic tetrapeptides targeting the testosterone binding site of ZIP9 stimulate myotube formation in myogenic cells and mineralization in osteogenic cells. Viveka Nand Malviya, Ahmed Bulldan, Raffael C. Wende, Marie-Louise Möller, Peter R. Schreiner and Georgios Scheiner-Bobis
Biology 2022, 11,19. DOI: 10.3390/biology11010019.ZIP9 is a recently identified membrane-bound androgen receptor of physiological significance that may mediate certain physiological responses to androgens. Using in silico methods, six tetrapeptides with the best docking properties at the testosterone binding site of ZIP9 were synthesized and further investigated. All tetrapeptides displaced T-BSA-FITC, a membrane-impermeable testosterone analog, from the surface of mouse myogenic L6 cells that express ZIP9 but not the classical androgen receptor (AR). Silencing the expression of ZIP9 with siRNA prevented this labeling. All tetrapeptides were found to be pro-androgenic; in L6 cells they stimulated the expression of myogenin, triggered activation of focal adhesion kinase, and prompted the fusion of L6 myocytes to syncytial myotubes. In human osteoblastic SAOS-2 cells that express AR and ZIP9, they reduced the expression of alkaline phosphatase and stimulated mineralization. These latter effects were prevented by silencing ZIP9 expression, indicating that the osteoblast/osteocyte conversion is exclusively mediated through ZIP9. Our results demonstrate that the synthetic tetrapeptides, by acting as ZIP9-specific androgens, have the potential to replace testosterone or testosterone analogs in the treatment of bone- or muscle-related disorders by circumventing the undesirable effects mediated through the classical AR.

DMAP immobilized on porous silica particles and monoliths for the esterification of phenylethanol in continuous flow. Julia S. Schulze, Raoul D. Brand, Joachim G.C. Hering, Peter R. Schreiner and Bernd M. Smarsly
ChemCatChem 2022, 14, e202101845. DOI: 10.1002/cctc.202101845.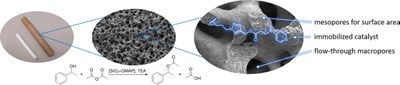
We report the immobilization of 4-dimethylaminopyridine (DMAP), a versatile organocatalyst for sterically demanding esterifications, on mesoporous silica particles and macro-mesoporous silica monoliths, both possessing optimized properties for continuous flow synthesis. An alkyne-functionalized DMAP derivative was immobilized via click chemistry; the materials were characterized by physisorption analysis, diffuse reflectance infrared Fourier transform spectroscopy (DRIFT) and elemental analysis. While silica particles were functionalized in batch and packed into a packed-bed reactor, monoliths were cladded with a polyether ether ketone (PEEK) tube after sol-gel synthesis and functionalized in a circulating flow process. Samples with three different catalyst loadings were prepared, in order to study the impact of the catalyst amount on the mesopore space as well as the catalytic performance. In continuous flow experiments, complete conversion of 1-phenylethanol to phenylethylacetate was achieved with both materials and short contact times. Monoliths exhibited far lower pressures than packed bed reactors (7 bar at a flow rate of 1 mL min−1) and reached turnover rates up to 9.3x10−2 s−1, which is almost twice as high as a comparable batch experiment. The absence of diffusion limitations in monoliths made investigations on reaction kinetics with microkinetics-dominated experiments possible. This study demonstrates that all properties needed for a successful transfer of immobilized organocatalysts to sophisticated flow syntheses with complex organocatalysts can be met with functionalized meso-macroporous monoliths.
Site-Selective Acylation of Pyranosides with Oligopeptide Catalysts. Alexander Seitz, Raffael C. Wende, Emily Roesner, Dominik Niedek, Christopher Topp, Avene C. Colgan, Eoghan M. McGarrigle and Peter R. Schreiner
J. Org. Chem. 2021, 86, 3907–3922. DOI: 10.1021/acs.joc.0c02772. Highlighted in B. List and B. Mitschke Synfacts 2021, 17, 698. DOI: 10.1055/s-0040-1706203Herein, we report the oligopeptide-catalyzed site-selective acylation of partially protected monosaccharides. We identified catalysts that invert site-selectivity compared to N-methylimidazole, which was used to determine the intrinsic reactivity, for 4,6-O-protected glucopyranosides (trans-diols) as well as 4,6-O-protected mannopyranosides (cis-diols). The reaction yields up to 81% of the inherently unfavored 2-O-acetylated products with selectivities up to 15:1 using mild reaction conditions. We also determined the influence of protecting groups on the reaction and demonstrate that our protocol is suitable for one-pot reactions with multiple consecutive protection steps.
Breaking the Symmetry of a meso-Compound by Isotopic Substitution: Synthesis and Stereochemical Assignment of Monodeuterated cis-Perhydroazulene. Fumito Saito, Dennis Gerbig, Jonathan Becker, and Peter R. Schreiner
Org. Lett. 2021, 23, 113–117. DOI: 10.1021/acs.orglett.0c03795We report the synthesis and absolute configuration of monodeuterated cis-perhydroazulene (d1-1), which is a rare example of an isotopically chiral hydrocarbon whose synthesis and stereochemical analysis are known to be particularly difficult. The synthesis features nickel-boride-catalyzed deuteration that allowed formation of the diastereomerically pure cis-fused bicyclic system in d1-1. The vibrational circular dichroism results are in excellent agreement with the computed spectrum at ωB97XD/aug-cc-pVTZ, allowing unambiguous assignment of the absolute configuration of d1-1.

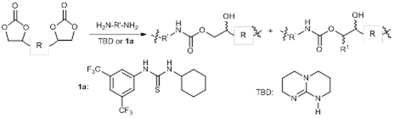
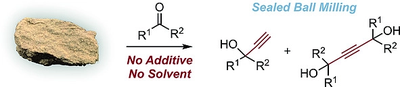
Direct Exploitation of the Ethynyl Moiety in Calcium Carbide Through Sealed Ball Milling. Abolfazl Hosseini and Peter R. Schreiner
Eur. J. Org. Chem. 2020, 28, 4339–4346. DOI: 10.1002/ejoc.202000612Sealed ball milling provides a novel way of activation of calcium carbide. No additive is required to transfer the acetylide moiety to organic molecules. Various ketones and even aryl halides can thereby be ethynylated.
Ball milling of calcium carbide (CaC2) enables the reaction of its ethynyl moiety with organic electrophiles. This was realized simply by co‐milling CaC2 with organic substrates in a sealed jar without the need for an additive or a catalyst. Various ketones including those bearing α‐hydrogens were ethynylated in good yields at short reaction times. Aryl halides are also amenable substrates for this protocol as they furnish aryl ethynes through a benzyne intermediate. This method offers a practical and cheap alternative to the established procedures for introducing ethynyl functionalities.
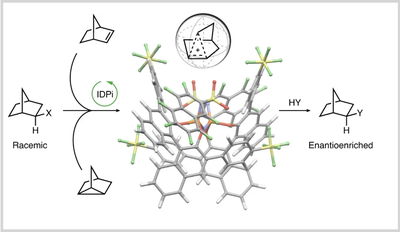
Catalytic enantiocontrol over a non-classical carbocation. Roberta Properzi, Philip S. J. Kaib, Markus Leutzsch, Raja Mitra, Gabriele Pupo, Chandra Kanta De, Lijuan Song, Peter R. Schreiner, Benjamin List
Nat. Chem. 2020, 12, 1174–1179. DOI: 10.1038/s41557-020-00558-1Carbocations can be categorized into classical carbenium ions and non-classical carbonium ions. These intermediates are ubiquitous in reactions of both fundamental and practical relevance, finding application in the petroleum industry as well as the discovery of new drugs and materials. Conveying stereochemical information to carbocations is therefore of interest to a range of chemical fields. While previous studies targeted systems proceeding through classical ions, enantiocontrol over their non-classical counterparts has remained unprecedented. Here we show that strong and confined chiral acids catalyse enantioselective reactions via the non-classical 2-norbornyl cation. This reactive intermediate is generated from structurally different precursors by leveraging the reactivity of various functional groups to ultimately deliver the same enantioenriched product. Our work demonstrates that tailored catalysts can act as suitable hosts for simple, non-functionalized carbocations via a network of non-covalent interactions. We anticipate that the methods described herein will provide catalytic accessibility to valuable carbocation systems.
TEMPO-functionalized mesoporous silica particles as heterogeneous oxidation catalysts in flow. Julia Schulze, Julia Migenda, M. Becker, Sören M. M. Schuler, Raffael C. Wende, Peter R. Schreiner, Bernd M. Smarsly
J. Mat. Chem. A 2020, 8, 4107–4117. DOI: 10.1039/c9ta12416k
Organocatalysts immobilized on inorganic porous substrates possess fundamental benefits, e.g., a high catalyst/reactant ratio, easy scalability as well as work-up, and continuous processing. Here we report the development of a heterogeneous (2,2,6,6-tetramethylpiperidin-1-yl)oxyl (TEMPO) catalyst immobilized on mesoporous SiO2 and demonstrate its feasibility in the oxidation of benzyl alcohol to benzaldehyde. Our catalyst system is based on commercially available mesoporous silica particles that are optimized for flow applications (LiChrospher® Si 100 from Merck). The transition from well-known silica materials like MCM-41 and SBA-15 to specialized high-performance materials such as the used LiChrospher® particles is of great value for getting closer to industrial applications on large scale. We functionalized the material by applying click-chemistry and employed a packed HPLC column for the investigation of the catalyst performance and stability in continuous flow. The material shows the best performance with low catalyst loadings. The catalytic activity can be improved significantly by conversion of the TEMPO radical to an oxoammonium salt prior to the reaction. The material is well suited for applications in continuous flow syntheses, as the spherical shape of the particles results in low back pressures. The organic catalyst produces yields up to 89% with a flow rate of 0.05 mL min−1. The mild reaction conditions allow the use of the material in multi-step reactions. This option was demonstrated by combining the TEMPO-functionalized column with an aminopropyl-functionalized column and performing a TEMPO-mediated oxidation followed by a Knoevenagel condensation in a continuous flow setup for the first time. Long-term tests and post-catalytic analysis show a previously neglected decomposition pathway of TEMPO due to the co-catalyst.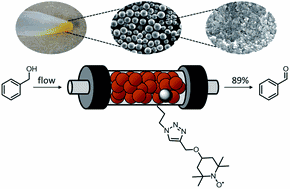
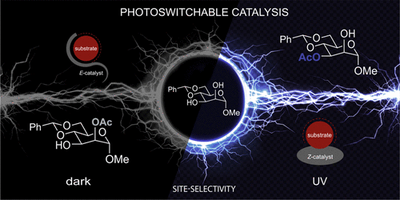
In Situ Switching of Site-Selectivity with Light in the Acetylation of Sugars with Azopeptide Catalysts. Dominik Niedek, Frederik R. Erb, Christopher Topp, Alexander Seitz, Raffael C. Wende, André K. Eckhardt, Jonas Kind, Dominik Herold, Christina M. Thiele and Peter R. Schreiner
J. Org. Chem. 2020, 85, 1835–1846.. DOI: 10.1021/acs.joc.9b01913We present a novel concept for the in situ control of site-selectivity of catalytic acetylations of partially protected sugars using light as external stimulus and oligopeptide catalysts equipped with an azobenzene moiety. The isomerizable azobenzene-peptide backbone defines the size and shape of the catalytic pocket, while the π-methyl-l-histidine (Pmh) moiety transfers the electrophile. Photoisomerization of the E- to the Z-azobenzene catalyst (monitored via NMR) with an LED (λ = 365 nm) drastically changes the chemical environment around the catalytically active Pmh moiety, so that the light-induced change in the catalyst shape alters site-selectivity. As a proof of principle, we employed (4,6-O-benzylidene)methyl-α-d-pyranosides, which provide a change in regioselectivity from 2:1 (E) to 1:5 (Z) for the monoacetylated products at room temperature. The validity of this new catalyst-design concept is further demonstrated with the regioselective acetylation of the natural product quercetin. In situ irradiation NMR spectroscopy was used to quantify photostationary states under continuous irradiation with UV light.
2-Unsubstituted Imidazole N-Oxides as Novel Precursors of Chiral 3-Alkoxyimidazol-2-ylidenes Derived from trans-1,2-Diaminocyclohexane and Other Chiral Amino Compounds. Grzegorz Mlostoń, Małgorzata Celeda, Marcin Jasinski, Katrzyna Urbaniak, Przemysław Boratyński, Peter R. Schreiner, Heinz Heimgartner
Molecules 2019, 24, 4398–4416. DOI: doi.org/10.3390/molecules24234398‘Desymmetrization’ of trans-1,2-diaminocyclohexane by treatment with α,ω-dihalogenated alkylation reagents leads to mono-NH2 derivatives (‘primary-tertiary diamines’). Upon reaction with formaldehyde, these products formed monomeric formaldimines. Subsequently, reactions of the formaldimines with α-hydroxyiminoketones led to the corresponding 2-unsubstituted imidazole N-oxide derivatives, which were used here as new substrates for the in situ generation of chiral imidazol-2-ylidenes. Upon O-selective benzylation, new chiral imidazolium salts were obtained, which were deprotonated by treatment with triethylamine in the presence of elemental sulfur. Under these conditions, the intermediate imidazol-2-ylidenes were trapped by elemental sulfur, yielding the corresponding chiral non-enolizable imidazole-2-thiones in good yields. Analogous reaction sequences, starting with imidazole N-oxides derived from enantiopure primary amines, amino alcohols, and amino acids, leading to the corresponding 3-alkoxyimidazole-2-thiones were also studied.
Syn-Dihydroxylation of Alkenes Using Sterically Demanding Cyclic Diacyl Peroxide. Afsaneh Pilevar, Abolfazl Hosseini and Peter R. Schreiner
J. Org. Chem. 2019, 84,12377–12386. DOI: 10.1021/acs.joc.9b01748
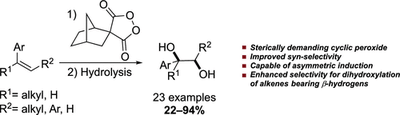
The syn-dihydroxylation of alkenes is a highly valuable reaction in organic synthesis. Cyclic acyl peroxides (CAPs) have emerged recently as promising candidates to replace the commonly employed toxic metals for this purpose. Here, we demonstrate that the structurally demanding cyclic peroxide spiro[bicyclo[2.2.1]heptane-2,4′-[1,2]dioxolane]-3′,5′-dione (P4) can be effectively used for the syn-dihydroxylation of alkenes. Reagent P4 also shows an improved selectivity for dihydroxylation of alkenes bearing β-hydrogens as compared to other CAPs, where both diol and allyl alcohol products compete with each other. Furthermore, the use of enantiopure P4 (labeled P4′) demonstrates the potential of P4′ for a metal-free asymmetric syn-dihydroxylation of alkenes.
Synthesis of Exclusively 4-Substituted β-Lactams through the Kinugasa Reaction Utilizing Calcium Carbide. Abolfazl Hosseini and Peter R. Schreiner
Org. Lett. 2019, 21, 3746-3749. DOI: 10.1021/acs.orglett.9b01192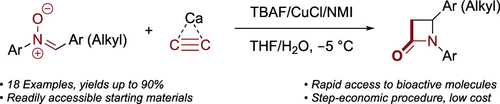
A new Kinugasa reaction protocol has been elaborated for the one-pot synthesis of 4-substituted β-lactams utilizing calcium carbide and nitrone derivatives. Calcium carbide is thereby activated by TBAF·3H2O in the presence of CuCl/NMI. The ease of synthesis and use of inexpensive chemicals provides rapid access of practical quantities of β-lactams exclusively substituted at position 4.
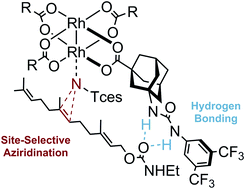
Site-Selective Nitrenoid Insertions Utilizing Postfunctionalized Bifunctional Rhodium(II) Catalysts. Jan-Philipp Berndt, Yevhenii Radchenko, Jonathan Becker, Christian Logemann, Dhaka R. Bhandari, Radim Hrdina and Peter R. Schreiner
Chem. Sci. 2019, 10, 3324–3329. DOI: 10.1039/C8SC05733HWe report a new strategy for the preparation of dirhodium(II) complexes with the general formula Rh2(A)4 that allows the isolation of a dirhodium tetracarboxylate complex with a free amino group available for postfunctionalization. The postfunctionalization of this complex enables the incorporation of a variety of functional groups, including double and triple bonds as well as nucleophilic moieties, thus paving the way to new classes of polymeric as well as bifunctional catalysts, and polymetallic complexes. Furthermore, we demonstrate that a urea containing dirhodium(II) complex enables site-selective nitrenoid insertions by remote hydrogen bonding control.
Selective Phthalimido-N-oxyl (PINO) Catalyzed C−H Cyanation of Adamantane Derivatives. Jan-Philipp Berndt, Frederik R. Erb, Lukas Ochmann, Jaqueline Beppler and Peter R. Schreiner
Synlett 2019, 30, 493–498. DOI: 10.1055/s-0037-1610403. Highlight: Pearl Issue
We present a new method for the selective C(sp3)–H cyanation of adamantane derivatives with PINO as the hydrogen abstracting reagent. A cyano radical is thereby transferred from p -toluenesulfonyl cyanide, allowing the cyanation of adamantane derivatives in up to 71% yield. The protocol presents a novel way to orthogonally functionalized adamantanes that are otherwise difficult to prepare. Mechanistic studies support the hypothesis of a radical pathway.
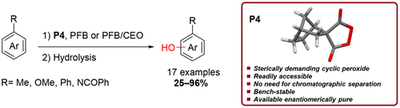
Tuning the Reactivity of Peroxo-Anhydrides for Aromatic C–H Bond Oxidation. Afsaneh Pilevar, Abolfazl Hosseini, Marina Šekutor, Heike Hausmann, Jonathan Becker, Kevin Turke and Peter R. Schreiner
J. Org. Chem. 2018, 83, 10070–10079. DOI: 10.1021/acs.joc.8b01392Phenol moieties are key structural motifs in many areas of chemical research from polymers to pharmaceuticals. Herein, we report on the design and use of a structurally demanding cyclic peroxide (spiro[bicyclo[2.2.1]heptane-2,4′-[1,2]dioxolane]-3′,5′-dione, P4) for the direct hydroxylation of aromatic substrates. The new peroxide benefits from high thermal stability and can be synthesized from readily available starting materials. The aromatic C–H oxidation using P4 exhibits generally good yields (up to 96%) and appreciable regioselectivities.
Catalytic Halogen Bond Activation in the Benzylic C–H Bond Iodination with Iodohydantoins. Sascha H. Combe, Abolfazl Hosseini, Henrik Quanz, Heike Hausmann and Peter R. Schreiner
Org. Lett. 2017, 19, 6156–6159. DOI: 10.1021/acs.orglett.7b03034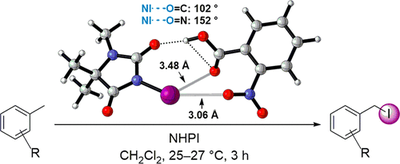
This letter presents the side-chain iodination of electron-deficient benzylic hydrocarbons at rt using N-hydroxyphthalimide (NHPI) as radical initiator and 1,3-diiodo-5,5-dimethylhydantoin and 3-iodo-1,5,5-trimethylhydantoin (3-ITMH) as iodine source. Addition of a carboxylic acid increased the reactivity due to complex formation with and activation of 3-ITMH by proton transfer and halogen bond formation. No SEAr reactions were observed under the employed reaction conditions. Our method enables convenient product isolation and gives 50–72% yields of isolated products.
Aliphatic C–H Bond Iodination by a N-Iodoamide and Isolation of an Elusive N-Amidyl Radical. Alexander Artaryan, Artur Mardyukov, Kseniya Kulbitski, Idan Avigdori, Gennady A. Nisnevich, Peter R. Schreiner and Mark Gandelman
J. Org. Chem. 2017, 82, 7093–7100. DOI: 10.1021/acs.joc.7b00557.Highlights: a) Nitrogen-based radical conquers challenging iodoalkane synthesis. Chem. Eng. News 2017, 95 (21), 9; b) ACS Editor’s Choice (open access); c) JOC cover page.
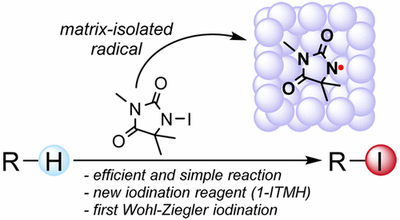
Contrary to C–H chlorination and bromination, the direct iodination of alkanes represents a great challenge. We reveal a new N-iodoamide that is capable of a direct and efficient C–H bond iodination of various cyclic and acyclic alkanes providing iodoalkanes in good yields. This is the first use of N-iodoamide for C–H bond iodination. The method also works well for benzylic C–H bonds, thereby constituting the missing version of the Wohl–Ziegler iodination reaction. Mechanistic details were elucidated by DFT computations, and the N-centered radical derived from the used N-iodoamide, which is the key intermediate in this process, was matrix-isolated in a solid argon matrix and characterized by UV–vis as well as IR spectroscopy.
Calcium Carbide Catalytically Activated with Tetra-n-butyl Ammonium Fluoride for Sonogashira Cross Coupling Reactions. Abolfazl Hosseini, Afsaneh Pilevar, Eimear Hogan and Peter R. Schreiner
Org. Biomol. Chem. 2017, 15, 6800–6807. DOI: 10.1039/C7OB01334EWe report a novel method for the direct synthesis of mono- and bis-arylated alkynes utilizing catalytically activated CaC2 as the alkyne component. This fluoride-activated cross coupling reaction provides advantages over existing methods regarding operational simplicity, use of readily available starting materials, and low cost.
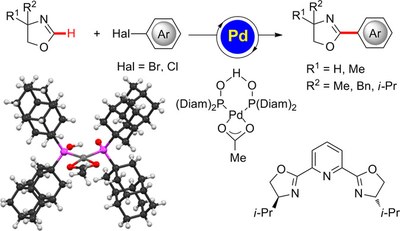
Secondary Phosphine Oxide-Palladium Catalyst for C–H (Hetero)Arylations: Efficient Access to Pybox Ligands. Debasish Ghorai, Valentin Müller, Helena Keil, Dietmar Stalke, Giuseppe Zanoni, Boryslav A. Tkachenko, Peter R. Schreiner, Lutz Ackermann
Adv. Synth. Cat. 2017, 359, 3137–3141. DOI: 10.1002/adsc.201700663. Highlight: inside cover.C–H arylations of oxazolines were accomplished with a well-defined palladium catalyst derived from a secondary bisdiamantyl phosphine oxide. The single-component secondary phosphine oxide (SPO)-palladium complex enabled C–H activations with aryl bromides and challenging aryl chlorides in the absence of directing groups, setting the stage for the step-economical synthesis of pybox ligands under racemization-free reaction conditions.
Mild Aliphatic and Benzylic C–H Bond Chlorination Using Trichloroisocyanuric Acid (TCCA). Sascha H. Combe, Abolfazl Hosseini, Alejandro Parra and Peter R. Schreiner*
J. Org. Chem. 2017, 82, 2407–2413. DOI: 10.1021/acs.joc.6b02829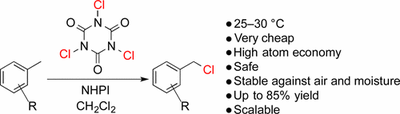
We present the controlled monochlorination of aliphatic and benzylic hydrocarbons with only 1 equiv of substrate at 25–30 °C using N-hydroxyphthalimide (NHPI) as radical initiator and commercially available trichloroisocyanuric acid (TCCA) as the chlorine source. Catalytic amounts of CBr4 reduced the reaction times considerably due to the formation of chain-carrying ·CBr3 radicals. Benzylic C–H chlorination affords moderate to good yields for arenes carrying electron-withdrawing (50–85%) or weakly electron-donating groups (31–73%); cyclic aliphatic substrates provide low yields (24–38%). The products could be synthesized on a gram scale followed by simple purification via distillation. We report the first direct side-chain chlorination of 3-methylbenzoate affording methyl 3-(chloromethyl)benzoate, which is an important building block for the synthesis of vasodilator taprostene.
Synthesis of Enantioenriched Phthalide and Isoindolinone Derivatives from 2-Formylbenzoic Acid. Dominik Niedek, Sören M. M. Schuler, Christian Eschmann, Raffael C. Wende, Alexander Seitz, Felix Keul and Peter R. Schreiner*
Synthesis 2016, 48, 371–382. DOI: 10.1055/s-0036-1589404Transformations of 2-formylbenzoic acid provide direct access to a series of heterocyclic organic

Urea- and thiourea-catalyzed aminolysis of carbonates. M. Blain, Honman Yau, L. Jean-Gérard, R. Auvergne, D. Benazet, Peter R. Schreiner, S. Caillol and Bruno Andrioletti
ChemSusChem 2016, 9, 2269-2272. DOI:The Enantioselective Dakin-West Reaction. Raffael C. Wende, Alexander Seitz, Dominik Niedek, Sören M. M. Schuler, Christine Hofmann, Jonathan Becker, and Peter R. Schreiner
Angew. Chem. Int. Ed. 2016, 55, 2719–2723. DOI: 10.1002/anie.201509863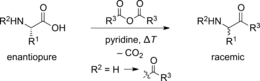
Here we report the development of the first enantioselective Dakin–West reaction, yielding α-acetamido methylketones with up to 58 % ee with good yields. Two of the obtained products were recrystallized once to achieve up to 84 % ee. The employed methylimidazole-containing oligopeptides catalyze both the acetylation of the azlactone intermediate and the terminal enantioselective decarboxylative protonation. We propose a dispersion-controlled reaction path that determines the asymmetric reprotonation of the intermediate enolate after the decarboxylation.
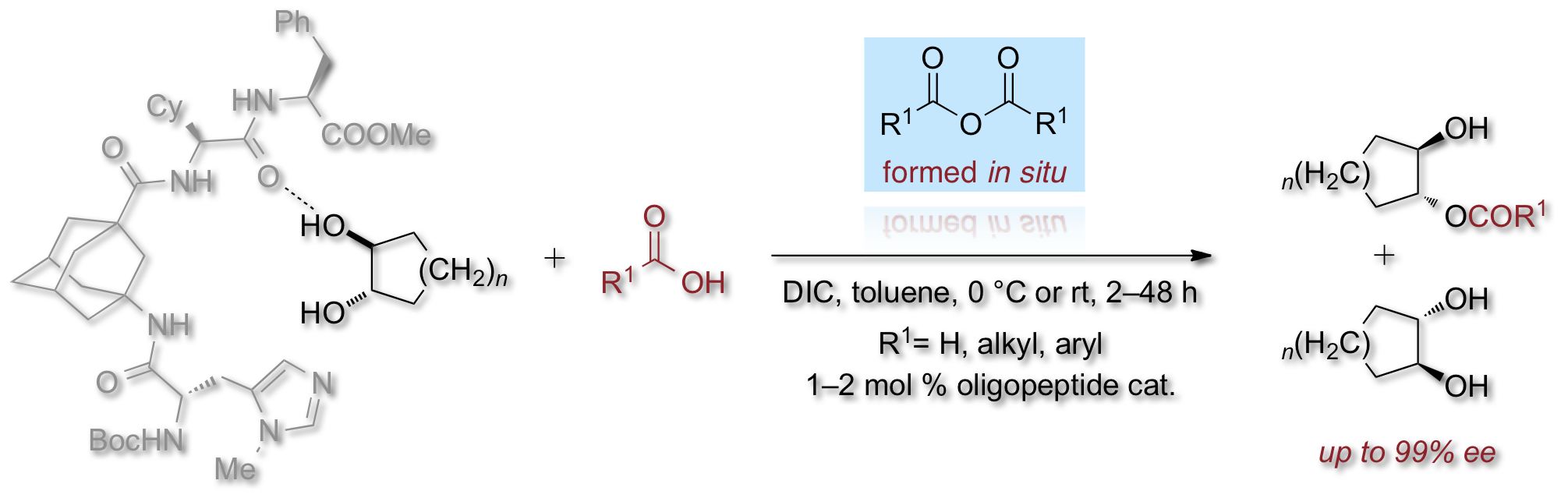
Functionality, Effectiveness, and Mechanistic Evaluation of a Multicatalyst-Promoted Reaction Sequence by Electrospray Ionization Mass Spectrometry. M. Wasim Alachraf, Raffael C. Wende, Sören M. M. Schuler, Peter R. Schreiner, and Wolfgang Schrader
Chem. Eur. J. 2015, 21, 16203-16208. DOI: 10.1002/chem.201502640
A multicatalytic three-step reaction consisting of epoxidation, hydrolysis, and enantioselective monoacylation of cyclohexene was studied by using mass spectrometry (MS). The reaction sequence was carried out in a one-pot reaction using a multicatalyst. All reaction steps were thoroughly analyzed by electrospray ionization (ESI) MS (and MS/MS), as well as high-resolution MS for structure elucidation. These studies allow us to shed light on the individual mode of action of each catalytic moiety. Thus, we find that under the epoxidation conditions, the catalytically active N-methyl imidazole for the terminal acylation step is partially deactivated through oxidation. This observation helps to explain the lower efficiency of the catalyst in the last step compared to the monoacylation performed separately. All reactive intermediates and products of the reaction sequence, as well as of the side-reactions, were monitored, and we present a working mechanism of the reaction.
Fluoride-assisted activation of calcium carbide: A simple and efficient method for the ethynylation of aldehydes and ketones. Abolfazl Hosseini, Daniel Seidel, Andreas Miska and Peter R. Schreiner
Org. Lett. 2015, 17, 2808–2811. DOI: 10.1021/acs.orglett.5b01219
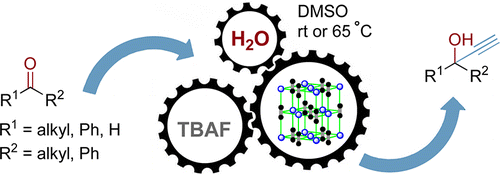 The fluoride-assisted ethynylation of ketones and aldehydes is described using commercially available calcium carbide with typically 5 mol % of TBAF·3H2O as the catalyst in DMSO. Activation of calcium carbide by fluoride is thought to generate an acetylide “ate”-complex that readily adds to carbonyl groups. Aliphatic aldehydes and ketones generally provide high yields, whereas aromatic carbonyls afford propargylic alcohols with moderate to good yields. The use of calcium carbide as a safe acetylide ion source along with economic amounts of TBAF·3H2O make this procedure a cheap and operationally simple method for the preparation of propargylic alcohols.
The fluoride-assisted ethynylation of ketones and aldehydes is described using commercially available calcium carbide with typically 5 mol % of TBAF·3H2O as the catalyst in DMSO. Activation of calcium carbide by fluoride is thought to generate an acetylide “ate”-complex that readily adds to carbonyl groups. Aliphatic aldehydes and ketones generally provide high yields, whereas aromatic carbonyls afford propargylic alcohols with moderate to good yields. The use of calcium carbide as a safe acetylide ion source along with economic amounts of TBAF·3H2O make this procedure a cheap and operationally simple method for the preparation of propargylic alcohols.A Dual-Catalysis Anion Binding Approach to the Kinetic Resolution of Amines: Insights into the Mechanism via a Combined Experimental and Computational Study. Nisha Mittal, Katharina M. Lippert, Chandra Kanta De, Eric G. Klauber, Thomas J. Emge, Peter R. Schreiner and Daniel Seidel
J. Am. Chem. Soc. 2015, 137, 5748–5758. DOI: 10.1021/jacs.5b00190
Racemic benzylic amines undergo kinetic resolution via benzoylation with benzoic anhydride in the
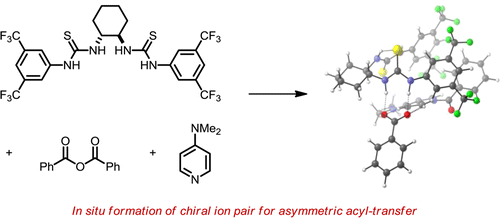 presence of a dual catalyst system consisting of a readily available amide-thiourea catalyst and 4-dimethylaminopyridine (DMAP). An evaluation of various experimental parameters was performed in order to derive a more detailed understanding of what renders this process selective. The catalyst’s aggregation behavior and anion-binding ability were evaluated in regard to their relevance for the catalytic process. Alternate scenarios, such as catalyst deprotonation or the in situ formation of a neutral chiral acylating reagent were ruled out. Detailed computational studies at the M06/6-31G(d,p) level of theory including solvent modeling utilizing a polarized continuum model provide additional insights into the nature of the ion pair and reveal a range of important secondary interactions that are responsible for efficient enantiodiscrimination.
presence of a dual catalyst system consisting of a readily available amide-thiourea catalyst and 4-dimethylaminopyridine (DMAP). An evaluation of various experimental parameters was performed in order to derive a more detailed understanding of what renders this process selective. The catalyst’s aggregation behavior and anion-binding ability were evaluated in regard to their relevance for the catalytic process. Alternate scenarios, such as catalyst deprotonation or the in situ formation of a neutral chiral acylating reagent were ruled out. Detailed computational studies at the M06/6-31G(d,p) level of theory including solvent modeling utilizing a polarized continuum model provide additional insights into the nature of the ion pair and reveal a range of important secondary interactions that are responsible for efficient enantiodiscrimination.Alcohol Cross-Coupling for the Kinetic Resolution of Diols via Oxidative Esterification. Christine Hofmann, Jan M. Schümann, and Peter R. Schreiner
J. Org. Chem. 2015, 80, 1972–1978. DOI: 10.1021/jo502670p. Open Access.
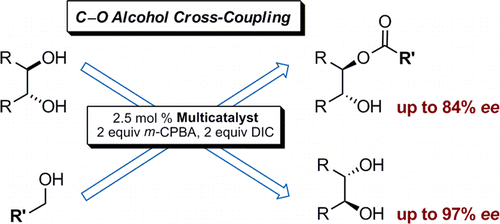
We present an organocatalytic C–O-bond cross-coupling strategy to kinetically resolve racemic diols with aromatic and aliphatic alcohols, yielding enantioenriched esters. This one-pot protocol utilizes an oligopeptide multicatalyst, m-CPBA as the oxidant, and N,N′-diisopropylcarbodiimide as the activating agent. Racemic acyclic diols as well as trans-cycloalkane-1,2-diols were kinetically resolved, achieving high selectivities and good yields for the products and recovered diols.
Spectroscopically Quantified Hydrogen-Bonding Strengths of Thioureas and their Catalytic Activity in Diels-Alder Reactions.
Alexander R. Nödling, Gergely Jakab, Peter R. Schreiner, and Gerhard Hilt
Eur. J. Org. Chem. 2014, 6394–6398. DOI: 10.1002/ejoc.201402871.
The hydrogen-bonding strength of a variety of commonly employed thiourea catalysts was quantified by using a trialkylphosphine oxide as a 31P NMR probe. Simple diarylthioureas and more complex bifunctional amine- and hydroxy-substituted thiourea derivatives were examined. Their catalytic activity was determined in a Diels–Alder reaction, and the obtained pseudo-first-order rate constants were correlated with the 31P NMR chemical shifts. A linear correlation between both variables was observed throughout the functionalized thioureas. The 31P NMR probe correlation fared better in comparison to a pKa correlation. Accordingly, the quantification presented herein by using a 31P NMR probe offers an elegant way to estimate the catalytic activity of thiourea catalysts in hydrogen-bond-activated reactions such as the Diels–Alder reaction.
En route to multicatalysis: kinetic resolution of trans-Cycloalkane-1,2-diols via oxidative esterification.
Christine Hofmann, Sören M. M. Schuler, Raffael C. Wende, Heike Hausmann, and Peter R. Schreiner*
Chem. Commun. 2014, 50, 1221–1223; DOI: 10.1039/C3CC48584F
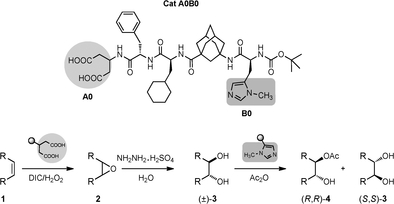
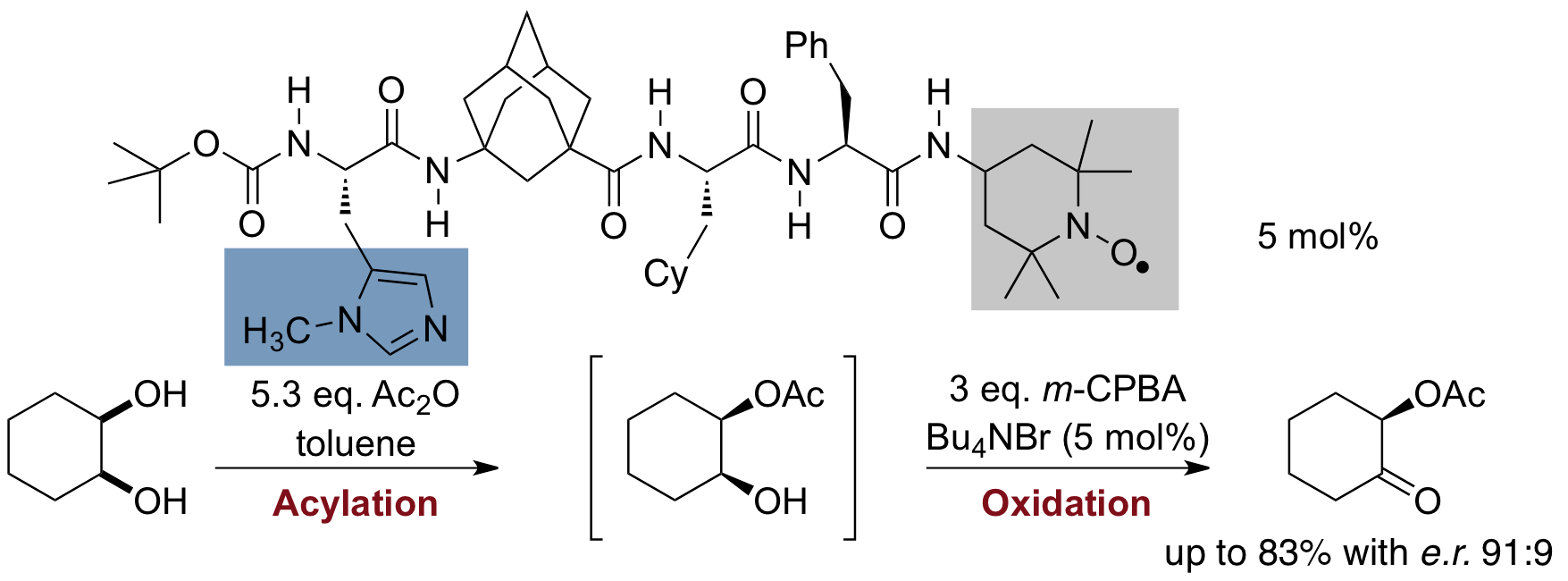
Lipophilic oligopeptides for chemo- and enantioselective acyl transfer onto alcohols. Christian E. Müller, Daniela Zell, Radim Hrdina, Raffael C. Wende, Lukas Wanka, Sören M. M. Schuler, and Peter R. Schreiner* J. Org. Chem. 2013, 78, 8465–8484. DOI: 10.1021/jo401195c
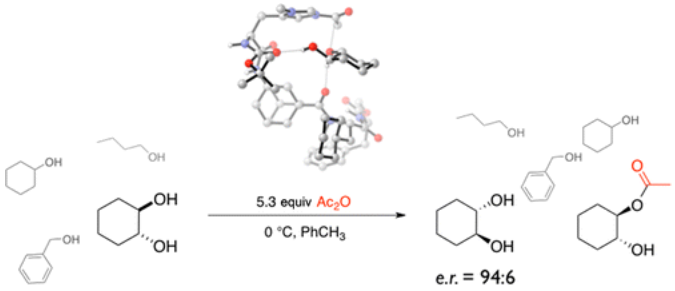
Inspired by the extraordinary selectivities of acylases, we envisioned the use of lipophilic oligopeptidic organocatalysts for the acylative kinetic resolution/desymmetrization of rac- and meso-cycloalkane-1,2-diols. Here we describe in a full account the discovery and development process from the theoretical concept to the final catalyst, including scope and limitations. Competition experiments with various alcohols and electrophiles show the full potential of the employed oligopeptides. Additionally, we utilized NMR and IR-spectroscopic methods as well as computations to shed light on the factors responsible for the selectivity. The catalyst system can be readily modified to a multicatalyst by adding other catalytically active amino acids to the peptide backbone, enabling the stereoselective one-pot synthesis of complex molecules from simple starting materials.
Mild and selective organocatalytic iodination of activated aromatic compounds. Gergely Jakab, Abolfazl Hosseini, Heike Hausmann, and Peter R. Schreiner*
Synthesis 2013, 45, 1635–1640. DOI: 10.1055/s-0033-1338468.
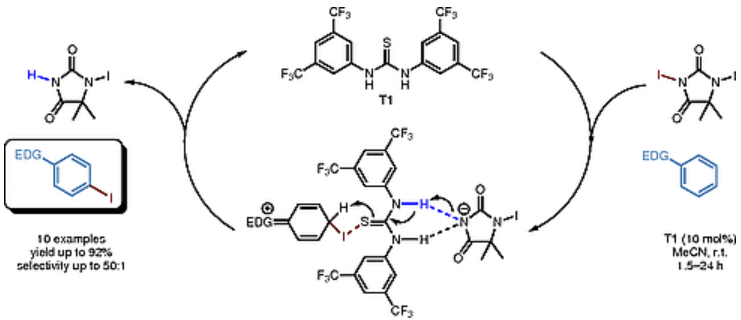
We describe an organocatalytic iodination of activated aromatic compounds using 1,3-diiodo-5,5-dimethylhydantoin (DIH) as the iodine source with thiourea catalysts in acetonitrile. The protocol is applicable to a number of aromatic substrates with significantly different steric and electronic properties. The iodination is generally highly regioselective and provides high yields of isolated products. NMR kinetic investigations conducted in THF-d8 indicate the role of sulfur in the thiourea motif as a nucleophile that is assisted by H-bonding in the key steps of the reaction.
Structure Analysis of Substrate-Catalyst Complexes in Mixtures with Ultrafast Two-Dimensional Infrared Spectroscopy. Andreas T. Messmer, Katharina M. Lippert, Peter R. Schreiner, and Jens Bredenbeck PCCP 2013, 15, 1509–1517. DOI: 10.1039/c2cp42863f.
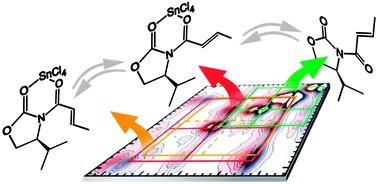
Ultrafast Two-Dimensional Infrared Spectroscopy Resolves the Conformational Change of an Evans Auxiliary Induced by Mg(ClO4)2.
Andreas T. Messmer, Sabrina Steinwand, Katharina M. Lippert, Peter R. Schreiner, and Jens Bredenbeck
J. Org. Chem. 2012, 77, 11091–11095. DOI: 10.1021/jo302160s.
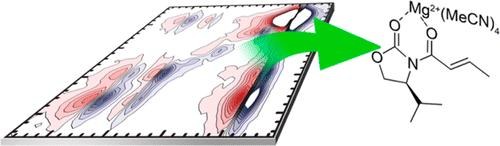
Structure determination of reactive species is a key step in understanding reaction mechanisms. We demonstrate the application of polarization-dependent two-dimensional infrared spectroscopy (P2D-IR) as a powerful tool combining structure resolution with ultrafast time resolution. We apply this technique to investigate the substrate–catalyst complexes in a Lewis acid catalyzed Diels–Alder reaction. Using Mg(ClO4)2 as a Lewis acid, we found that an additional complex besides the chelate typically postulated as reactive species forms. Experimental access to this new species leads to a deeper understanding of the observed selectivities for the Diels–Alder reaction catalyzed by Lewis acids. Our findings are supported by density functional computations at the M06/6-31+G(d,p) level, including solvent corrections.
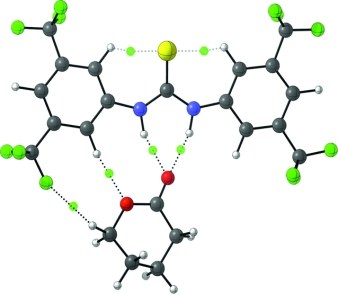
Hydrogen-Bonding Thiourea Organocatalysis: The Privileged 3,5-Bis(trifluoromethyl)phenyl Group.
Katharina M. Lippert, Kira Hof, Dennis Gerbig, David Ley, Heike Hausmann, Sabine Guenther, and Peter R. Schreiner Eur. J. Org. Chem. 2012, 5919–5927. DOI: 10.1002/ejoc.201200739.
Listed as one of the 10 most accessed articles 9/2012.
(Thio)urea Organocatalyst Equilibrium Acidities in DMSO.
Gergely Jakab, Carlo Tancon, Zhiguo Zhang, Katharina M. Lippert, and Peter R. Schreiner*
Org. Lett. 2012, 14, 1724–1727. DOI: 10.1021/ol300307c.
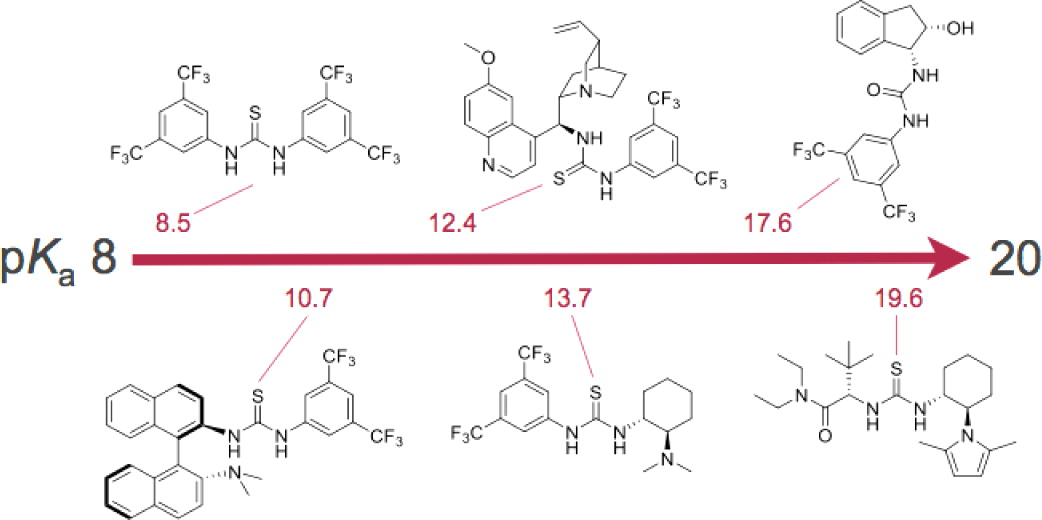
Listed as one of the 20 most accessed articles 2012.
Bordwell’s method of overlapping indicators was used to determine the pKa values of some of the most popular (thio)urea organocatalysts via UV spectrophotometric titrations. The incremental effect of CF3 groups on acidic strength was also investigated. The pKa’s are in the range of 8.5–19.6. The results may lead to a better understanding of noncovalent organocatalysis and may aid in future catalyst development.
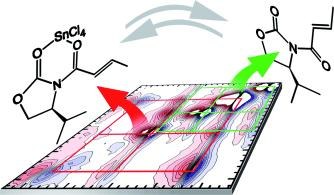
Two-dimensional Infrared Spectroscopy Reveals Structural Details of an Evans Auxiliary and its SnCl4 Complex. Andreas T. Messmer, Katharina M. Lippert, Sabrina Steinwand, Eliza-Beth W. Lerch, Kira Hof, David Ley, Dennis Gerbig, Heike Hausmann, Peter R. Schreiner, and Jens Bredenbeck Chem. Eur. J. 2012, 18, 14989–14995. DOI: 10.1002/chem.201201583.
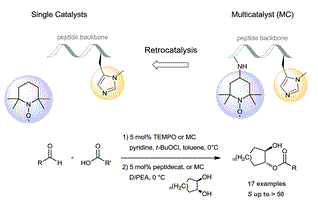
Evolution of Asymmetric Organocatalysis: Multi- and Retrocatalysis. Raffael C. Wende and Peter R. Schreiner* Green Chem. 2012, 14, 1821–1849. DOI: 10.1039/C2GC35160A. Highlights: a) Front cover of this issue; b) listed as one of the most accessed articles 04/2012 [http://pubs.rsc.org/en/journals/journalissues/gc#!mostreadarticles].

Enantiomerically Enriched trans-Diols from Alkenes in One Pot: A Multicatalyst Approach.
Radim Hrdina, Christian E. Müller, Raffael C. Wende, Lukas Wanka, and Peter R. Schreiner*
Chem. Commun. 2012, 48, 2498–2500, DOI: 10.1039/C2CC17435A
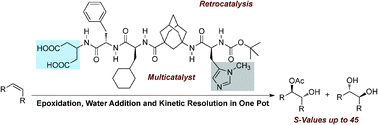
Multicatalysts consisting of non-natural oligopeptides with distinctly different catalytic moieties create molecular complexity in a multistep one-pot sequence starting from simple alkenes yielding highly enantiomerically enriched trans-diols.
Cooperative Thiourea-Brønsted Acid Organocatalysis: Direct Enantioselective Cyanosilylation of Aldehydes with TMSCN. Zhiguo Zhang, Katharina M. Lippert, Heike Hausmann, Mike Kotke, and Peter R. Schreiner*
J. Org. Chem. 2011, 76, 9764–9776, DOI: 10.1021/jo201864e
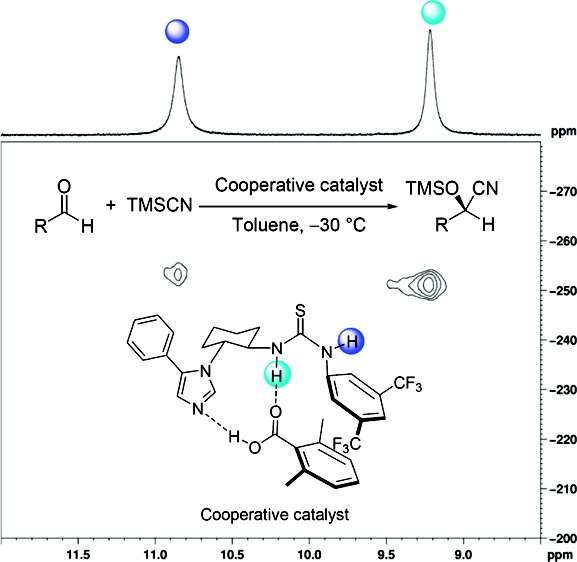 We report a new thiourea–Brønsted acid cooperative catalytic system for the enantioselective cyanosilylation of aldehydes with yields up to 90% and enantioselectivities up to 88%. The addition of an achiral acid was found to be crucial for high asymmetric induction. Mechanistic investigations using a combination of NMR, ESI-MS, and density functional theory computations (including solvent corrections) at the M06/6-31G(d,p) level of theory suggest that the key catalytic species results from the cooperative interaction of bifunctional thioureas and an achiral acid that form well-defined chiral hydrogen-bonding environments.
We report a new thiourea–Brønsted acid cooperative catalytic system for the enantioselective cyanosilylation of aldehydes with yields up to 90% and enantioselectivities up to 88%. The addition of an achiral acid was found to be crucial for high asymmetric induction. Mechanistic investigations using a combination of NMR, ESI-MS, and density functional theory computations (including solvent corrections) at the M06/6-31G(d,p) level of theory suggest that the key catalytic species results from the cooperative interaction of bifunctional thioureas and an achiral acid that form well-defined chiral hydrogen-bonding environments.Silicon-(Thio)urea Lewis-Acid Catalysis. Radim Hrdina, Christian E. Müller, Raffael C. Wende, Katharina M. Lippert, Mario Benassi, Bernhard Spengler, and Peter R. Schreiner*
J. Am. Chem. Soc. 2011, 133, 7624–7627.
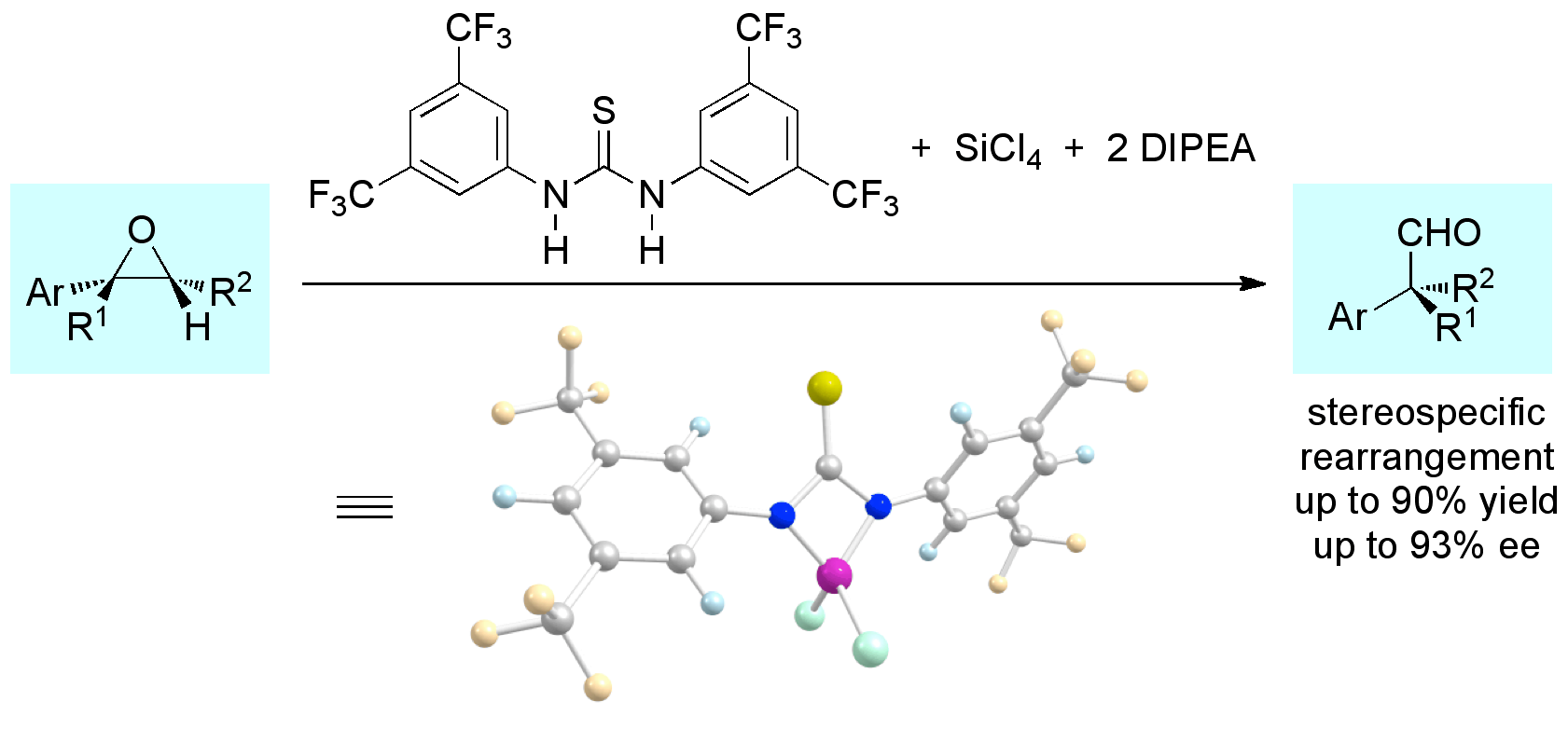
We present a new class of catalyst based on the combination of N,N´-diaryl-(thio)ureas and weak silicon Lewis acids (e.g., SiCl4). Such silicon-(thio)urea catalyst effectively catalyze the stereospecific rearrangement of epoxides to quaternary carbaldehydes.
A Multicatalyst System for the One-Pot Desymmetrization / Oxidation of meso-1,2-Alkane Diols. Christian E. Müller, Radim Hrdina, Raffael C. Wende, and Peter R. Schreiner* Chem. Eur. J. 2011, 17, 6309–6314.
Two is better than one: We demonstrate the viability of an organocatalytic reaction sequence along a short peptide backbone that carries two independent catalytic functionalities. This allows the rapid one-pot acylative desymmetrization and oxidation of meso-alkane-1,2-diols to the corresponding acetylated acetoins with good yields and enantioselectivities. The logical extension of this approach would be the generation of programmable oligopeptide sequences that assemble complex organic structures and even natural products in one pot.
Organocatalytic Enantioselective Acyl Transfer onto Racemic as well as meso Alcohols, Amines, and Thiols.Christian E. Müller and Peter R. Schreiner*
Angew. Chem. Int. Ed. 2011, 50, 6012–6042.
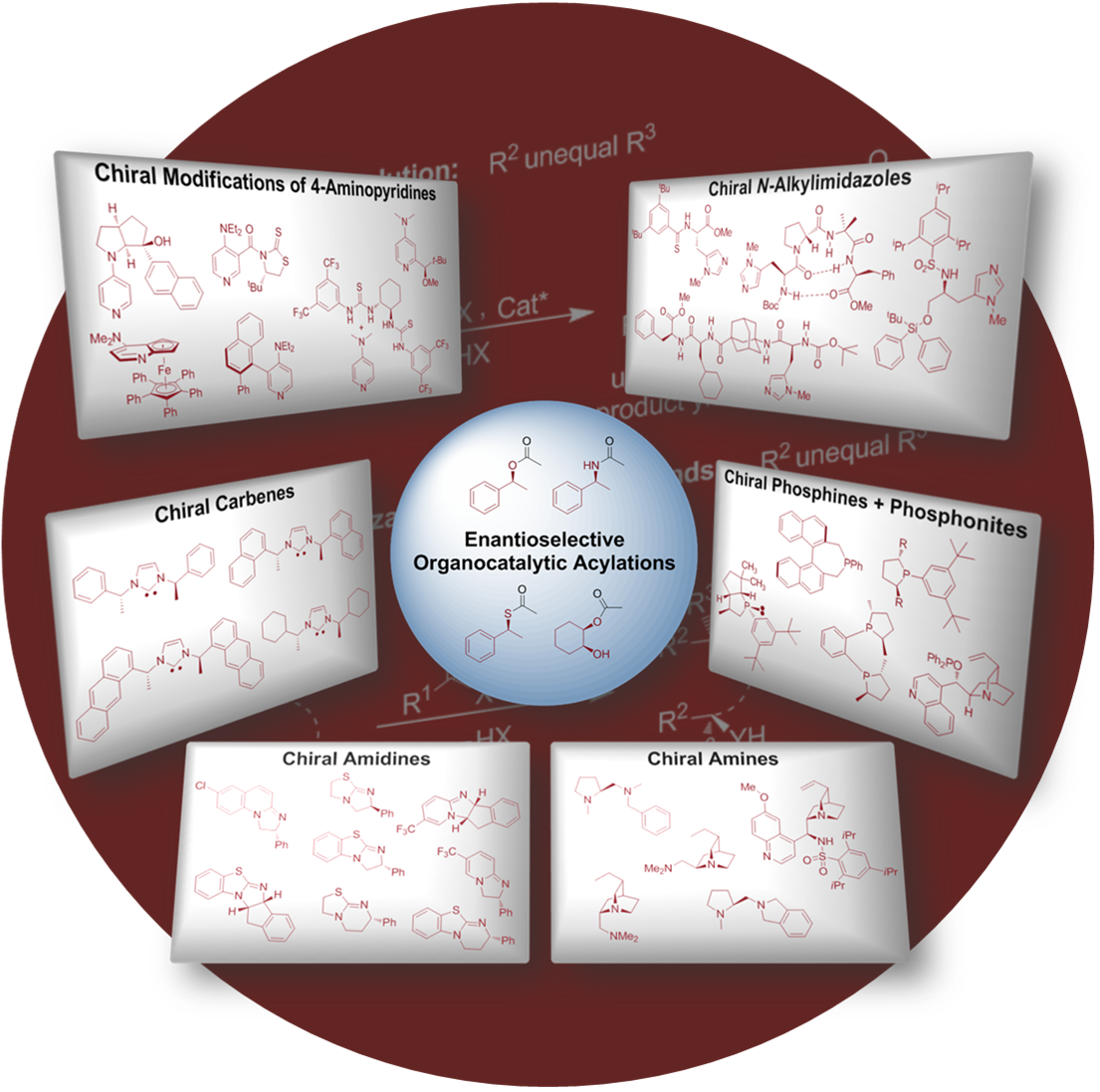
Acyl transfer is at the heart of functional group transfers utilized both in nature and in the chemical laboratory. Acylations are part of the natural assembly machinery for the generation of complex molecules and for biological energy transport. The recognition of covalent acyl-enzyme intermediates propelled both mechanistic studies as well as the development of biomimetic approaches. Consequently, chemists first used the tools of nature (e.g., employing enzymes and naturally occurring alkaloids as catalysts), but eventually developed a large variety of synthetic small molecules for selective acyl transfer. In contrast to nature, chemists utilize acylation reactions as a practical way for stereoselection and functional group protection. Indeed, the number of studies concerning acyl transfer in chemistry has significantly increased over the last 15 years. The present review examines and highlights these recent developments with the focus as given in the title.
Kinetic resolution of trans-cycloalkane-1,2-diols via Steglich Esterification. Radim Hrdina, Christian E. Müller, and Peter R. Schreiner
Chem. Commun. 2010, 46, 2689–2690.
We describe the efficient and highly enantioselective kinetic resolution of trans-cycloalkane-1,2-diols utilizing an enantio-selective Steglich reaction with a variety of carboxylic acids that form the corresponding anhydrides in situ.
Cooperativity Tames Reactive Catalysts. Peter R. Schreiner Science 2010, 327, 965–966.
When chemists use more than one catalyst to speed up a chemical reaction, it is almost always the case that overall increase in reaction rate is greater than that for either catalyst alone. For example, for acid-catalyzed reactions, combinations of "designer acids"—combinations of organic and inorganic molecules bearing acidic groups—can result in higher reactivity, selectivity, and versatility than the use of individual catalysts. However, there can be advantages to adding a second catalyst that actually slows down the rate of an already catalyzed reaction, especially if the goal is suppressing unwanted side reactions. This strategy is similar to that used by a coach of a relay team, who may pair a slower but sure-handed runner with a faster but less agile one. Speed is sacrificed to avoid losing by dropping the baton. Two catalysts that cooperate to control stereochemistry, can pick between different spatial arrangements of the same product. The initial catalyst is fast but produces an unwanted mixture of stereoisomers; adding the second catalyst suppresses one of these pathways and selects for the desired product. 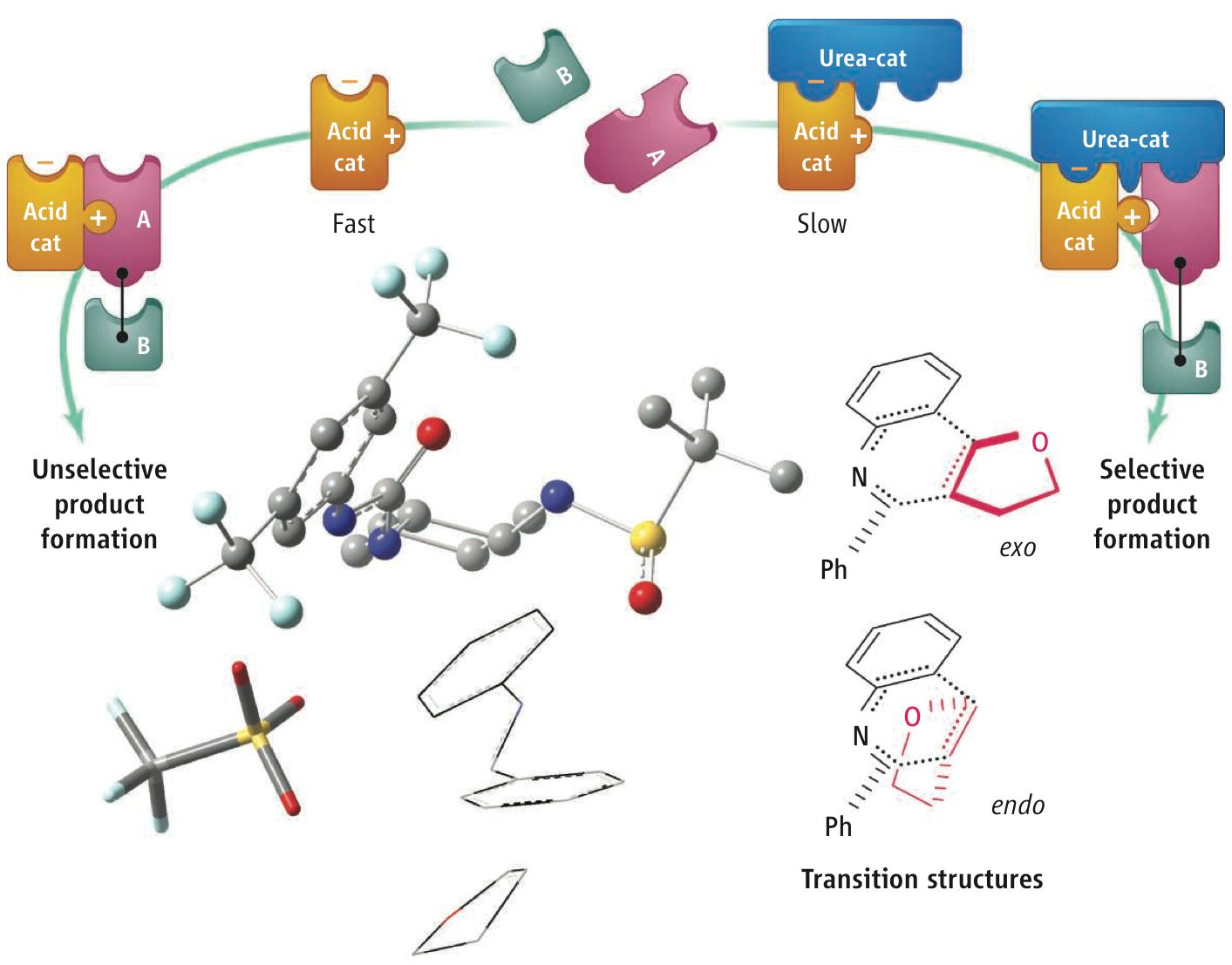
Asymmetric Transfer Hydrogenation of Ketimines with Trichlorosilane: Structural Studies.
Zhiguo Zhang, Parham Rooshenas, Heike Hausmann, and Peter R. Schreiner Synthesis 2009, 1531–1544.
We report structural and mechanistic studies on the organocatalytic asymmetric transfer hydrogenation of ketimines with trichlorosilane. Amines were obtained in good yields and moderate enantioselectivities. Both experiment and computation were utilized to provide an improved understanding of the mechanism. One-Pot Desymmetrization of meso-1,2-Hydrocarbon Diols through Acylation and Oxidation.Christian E. Müller, Daniela Zell, and Peter R. Schreiner Chem. Eur. J. 2009, 15, 9647–9650.Avoid racemization! Short lipophilic oligopeptides utilizing nucleophilic N-π-methyl histidine residues catalyze the desymmetrization of meso-1,2-diols with enantiomeric ratios of up to 94:6. Direct one-pot oxidation, which avoids the well-known racemization of the monoacylated product, directly leads to alpha-acetoxy ketones with enantiomeric ratios of up to 97:3 and 97% yield. 
Structural Analyses of N-Acetyl DMAP Salts. Volker Lutz, Jörg Glatthaar, Christian Würtele, Michael Serafin, Heike Hausmann, and Peter R. Schreiner Chem. Eur. J. 2009, 15, 8548.
Abstract. We studied the formation of several N-acetyl-DMAP salts (with Cl–, CH3COO–, and CF3COO– counterions), which are considered to be the catalytically active species in DMAP-catalyzed acetylation reactions of alcohols. Combined crystal structure analyses, variable temperature matrix IR and NMR spectroscopy as well as computational techniques at the UAHF-PCM-B3LYP/6-311+G(d,p)//B3LYP/6-31G(d) level were utilized to examine the structures and dynamics of salt formation. We see clear evidence for the formation of tight ion pairs that are stabilized by dynamic hydrogen-bonding interactions. In nonpolar solvents, the nucleophilicity of acetate in its N-acetyl-DMAP salt only allows a steady-state concentration smaller 1% at room temperature. Thus, we propose additional hydrogen bonding interactions with alcohols to be the key stabilization factor in subsequent acetylations.
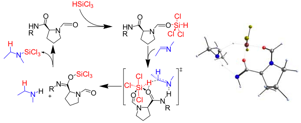
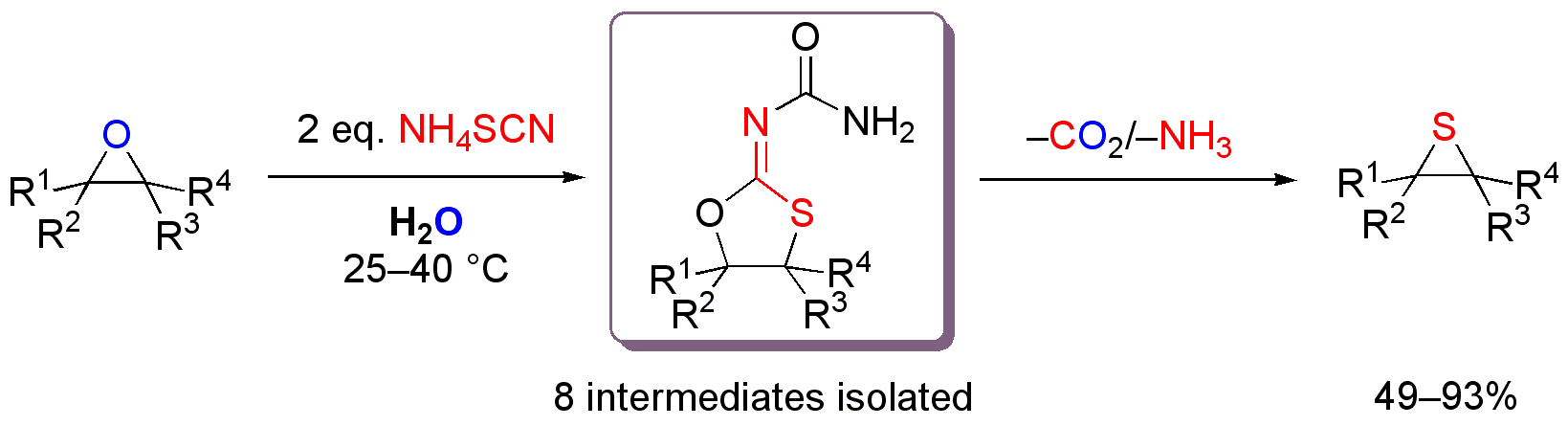
Isolation of the key intermediates in the catalyst free conversion of oxiranes to thiiranes in water at ambient temperature. Christian M. Kleiner, Luise Horst, Christian Würtele, Raffael Wende, and Peter R. Schreiner Org. Biomol. Chem. 2009, 7, 1397.Abstract. The highly practical conversion of oxiranes to thiiranes in water proceeds through (1,3-oxathiolan-2-ylidene)urea intermediates that could be fully characterized (X-ray) for the first time. Computational studies provide insights into the reaction mechanism.
(Thio)urea organocatalysis—What can be learnt from anion recognition?
Zhiguo Zhang and Peter R. Schreiner Chem. Soc. Rev. 2009, 38, 1187.
Abstract. The present critical review outlines the close relationship and mutual interplay between molecular recognition, active site considerations in enzyme catalysis involving anions, and organocatalysis utilizing explicit hydrogen bonding. These interconnections are generally not made although, as we demonstrate, they are quite apparent as exemplified with pertinent examples in the field of (thio)urea organocatalysis. Indeed, the concepts of anion binding or binding with negatively (partially) charged heteroatoms is key for designing new organocatalytic transformations. Utilizing anions through recognition with hydrogen-bonding organocatalysts is still in its infancy but bears great potential. In turn, the discovery and mechanistic elucidation of such reactions is likely to improve the understanding of enzyme active sites (108 references). 
Cooperative Bronsted Acid Type Organocatalysis: Alcoholysis of Styrene Oxides. Torsten Weil, Mike Kotke, Christian Kleiner, and Peter R. Schreiner Org. Lett. 2008, 10, 1513. Highlight: Benjamin List, Corinna Reisinger Synfacts 2008, 1513.
Abstract. We present a mild and efficient method for the completely regioselective alcoholysis of styrene oxides utilizing a cooperative Brønsted acid-type organocatalytic system comprised of mandelic acid (1 mol %) and N,N‘-bis-[3,5-bis-(trifluoromethyl)phenyl]-thiourea (1 mol %). Various styrene oxides are readily transformed into their corresponding β-alkoxy alcohols in good to excellent yields at full conversion. Simple aliphatic and sterically demanding, as well as unsaturated and acid-sensitive alcohols can be employed.
Enantioselective Kinetic Resolution of trans-cycloalkane-1,2-diols. Christian E. Müller, Lukas Wanka, Kevin Jewell, and Peter R. Schreiner Angew. Chem. 2008, 120, 6275.
Abstract. We have developed a peptide platform incorporating non-natural γ-Aminoadamantanecarboxylic Acids as scaffold that holds the catalytic site of a peptide and the centers governing recognition and stereochemistry in place. Our approach does not follow established oligopeptide catalyst design principles, which emphasize the formation of catalytically important secondary structures and showed to be highly successful and superior to enzyme catalysis in the kinetic resolution of trans-cycloalkane-1,2-diols.Thiourea Catalyzed Transfer Hydrogenation of Aldimines. Zhiguo Zhang and Peter R. Schreiner Synlett 2007, 1455. Highlight: Benjamin List, Subhas Chandra Pan Synfacts 2007, 988.
Abstract. The present letter reports on the thiourea-catalyzed transfer hydrogenation of imines through hydrogen-bonding activation with Hantzsch 1,4-dihydropyridine as the hydrogen source. A variety of aromatic as well as aliphatic aldimines can be reduced to give the respective amines under acid- and metal-free reaction conditions. 
Organocatalytic Biomimetic Reduction of Conjugated Nitroalkenes. Zhiguo Zhang and Peter R. Schreiner Synthesis, 2007, 2559.
Abstract. A thiourea catalyzed biomimetic reduction of conjugated nitroalkenes has been developed. Various aromatic and aliphatic conjugated nitroalkenes can be reduced to give the respective nitroalkanes with good yields under mild conditions. This protocol is not only practical, but may contribute to provide insight into the mechanisms of redox transformations in biological systems. 
Metal-free Organocatalysis Through Explicit Hydrogen Bonding Interactions. Peter R. Schreiner Chem. Soc. Rev. 2003, 32, 289.
Abstract. The metal(-ion)-free catalysis of organic reactions is a contemporary challenge that is just being taken up by chemists. Hence, this field is in its infancy and is briefly reviewed here, along with some rough guidelines and concepts for further catalyst development. Catalysis through explicit hydrogen bonding interactions offers attractive alternatives to metal (ion)-catalyzed reactions by combining supramolecular recognition with chemical transformations in an environmentally benign fashion. Although the catalytic rate accelerations relative to uncatalyzed reactions are often considerably less than for the metal (ion)-catalyzed variants, this need not be a disadvantage. Also, owing to weaker enthalpic binding interactions, product inhibition is rarely a problem and hydrogen bond additives are truly catalytic, even in water. 
Metal-Free, Noncovalent Catalysis of Diels-Alder Reactions by Neutral Hydrogen Bond Donors in Organic Solvents and in Water. A. Wittkopp, Peter R. Schreiner, Chem. Eur. J. 2003, 9, 407.
Abstract. We examined the catalytic activity of substituted thioureas in a series of Diels-Alder reactions and 1,3‑dipolar cycloadditions. The kinetic data reveal that the observed relative rate accelerations depend more on the thiourea substituents than on the reactants or solvent. Although the catalytic effectiveness is the strongest in non-coordinating, non-polar solvents such as cyclohexane, it is also present in highly coordinating polar solvents like water. In 1,3‑dipolar cycloadditions the thiourea catalysts demonstrate very moderate selectivity for reactions with inverse electron demand. Our experiments emphasize that both hydrophobic and polar interactions can co-exist, making these catalysts active even in highly coordinating solvents. This class of catalysts increases the reaction rates and endo-selectivities of Diels-Alder reactions, similar to weak Lewis acids, without concomitant product inhibition.
H-Bonding Additives Act Like Lewis Acid Catalysts.
Peter R. Schreiner, A. Wittkopp, Org. Lett. 2002, 4, 217.
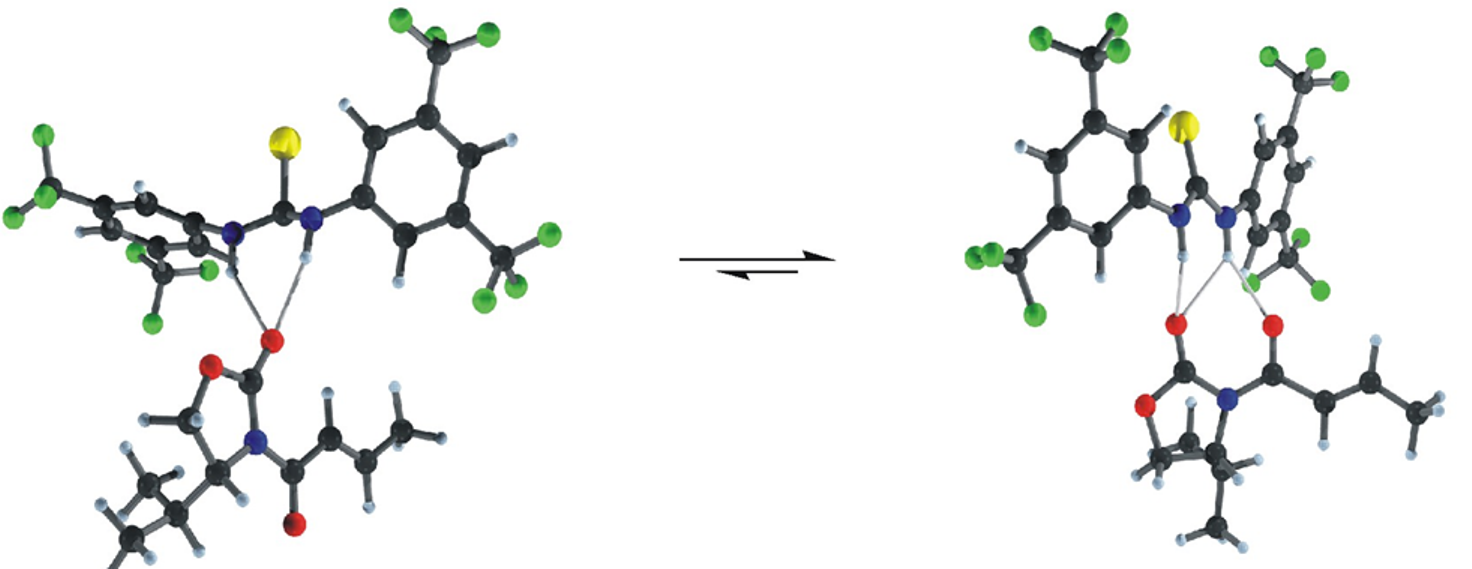
Abstract. A combination of NMR, IR, and ab initio techniques reveals the striking structural similarities of an exemplary N‑acyloxazolidinone in an H‑bonded complex with an N,N’-disubstituted electron-poor thiourea and the corresponding Lewis-acid complex. Although the H-bond association constant is lower than for the Lewis-acid adduct, Diels-Alder reactions are accelerated and stereochemically altered in a fashion similar to weak Lewis-acids.