Reactive Intermediates
Singlet carbenes incorporating a divalent carbon atom (R–C–R’) have grown from laboratory curiosities and theoreticians’ pet peeves into reagents in the growing field of stable (i.e., heterocyclic) carbene chemistry. Still, the experimental characterization of many other simple yet fundamentally important reactive species such as alkyl- or hydroxycarbenes, silylenes or sulfenes is hampered by their high reactivity or lack of precursors: Hydroxycarbenes, for instance, have been an unknown class of compounds until 2008, when our group reported the synthesis and characterization of hydroxymethylene (H–C–OH), whose preparation has been challenging organic chemists for more than 80 years. The reaction of hydroxycarbene with formaldehyde would be a source of simple sugars (the so-called “formose reaction” in the origin of life theory). Considerable efforts are ongoing to understand the formation and distribution of a wide variety of simple organics in extraterrestrial environments, and the examination of the structures and reactivities of prototypes such as various hydroxycarbenes, simple carboxylic acids or seemingly "exotic" heteroatom-containing compounds may also provide glimpses of the prebiotic earth.
Director: Univ.-Prof. Peter R. Schreiner
Co-workers:
Akkad Danho, Dr. Dennis Gerbig, Dr. Artur Mardyukov, Weiyu Qian, Dr. Nikolaos Vagkidis
Current work:
Hydroxy Mercapto Methylene: The Missing H2CSO Isomer
Markus Schauermann and Peter R. Schreiner
J. Phys. Chem. Lett.2022, 13, 3138–3142. DOI: 10.1021/acs.jpclett.2c00344.
We report the isolation of hydroxy mercapto methylene (HO–C̈–SH) under cryogenic conditions via pyrolysis of 2-ethoxy-2-thioxo-acetic acid. The two most stable carbene rotamers form via extrusion of ethylene and CO2 from this precursor. This donor-stabilized carbene represents a hitherto uncharacterized CH2SO species and the first spectroscopically characterized free mercapto carbene. CCSD(T)/cc-pVTZ computations support our findings.
On-Surface Synthesis and Visualization of Fleetingly Existent D3h-Cyclotriphosphazene (P3N3): of a Phosphorous-Nitrogen Analogue of Benzene.
Artur Mardyukov, Qigang Zhong, Ephrath Solel, Daniel Ebeling, Andre Schirmeisen and Peter R. Schreiner
Angew. Chem. Int. Ed. 2022, 61, revision re-submitted xx.yy.2022.
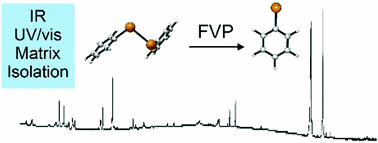
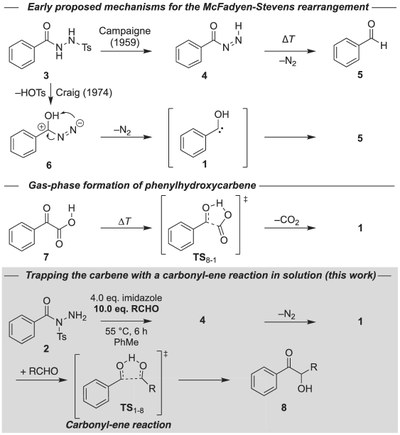
Generation and Reactivity of Phenylhydroxycarbenes in Solution.
Felix Keul, Artur Mardyukov and Peter R. Schreiner
J. Phys. Org. Chem. 2022, xx, yy–zz. in press. DOI: 10.1002/poc.4315.
We provide evidence for the first successful generation of phenylhydroxycarbene and 4-trifluoromethylphenylhydroxycarbene in solution. The carbene tautomers of the corresponding benzaldehyde derivatives had been prepared under cryogenic matrix-isolation conditions before but their reactivity, apart from a prototypical quantum mechanical tunneling [1,2]-H-shift reaction, had not been studied. Here our strategy is to employ suitable carbene precursors for the McFadyen–Stevens reaction, to generate the parent and the para-CF3-substituted phenylhydroxycarbenes, and to react them with benzaldehyde or acetone in a highly facile, allowed six-electron carbonyl-ene reaction toward the corresponding α-hydroxy ketones. Our findings are supported by computations at the DLPNO-CCSD(T)/cc-pVQZ//B3LYP/def2-TZVP level of theory.
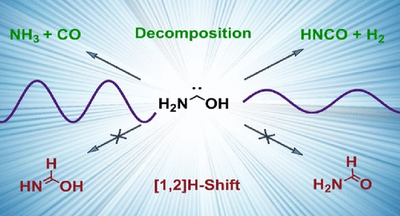
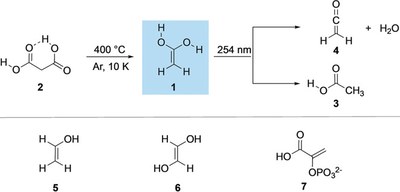
Aminohydroxymethylene (H2N–C̈–OH) – The Simplest Aminooxycarbene.
Bastian Bernhardt, Marcel Ruth, Hans Peter Reisenauer and Peter R. Schreiner
J. Phys. Chem. A 2021, 125, 7023–7028. DOI: 10.1021/acs.jpca.1c06151. Highlight: Featured in Chemviews August 13, 2021.
We generated and isolated hitherto unreported aminohydroxymethylene (1, aminohydroxycarbene) in solid Ar via pyrolysis of oxalic acid monoamide (2). Astrochemically relevant carbene 1 is persistent under cryogenic conditions and only decomposes to HNCO + H2 and NH3 + CO upon irradiation of the matrix at 254 nm. This photoreactivity is contrary to other hydroxycarbenes and aminomethylene, which undergo [1,2]H shifts to the corresponding carbonyls or imine. The experimental data are well supported by the results of CCSD(T)/cc-pVTZ and B3LYP/6-311++G(3df,3pd) computations.
Identification of a prismatic P3N3 molecule formed from electron irradiated phosphine-nitrogen ices.
Cheng Zhu, André K. Eckhardt, Sankhabrata Chandra, Peter R. Schreiner, Ralf I. Kaiser
Nat. Commun. 2021, 12,5467. DOI: 10.1038/s41467-021-25775-1. Highlight: Featured in Chem. Eng. News 2021, 99(35), x.
Polyhedral nitrogen containing molecules such as prismatic P3N3 - a hitherto elusive isovalent species of prismane (C6H6) - have attracted particular attention from the theoretical, physical, and synthetic chemistry communities. Here we report on the preparation of prismatic P3N3 [1,2,3-triaza-4,5,6-triphosphatetracyclo[2.2.0.02,6.03,5]hexane] by exposing phosphine (PH3) and nitrogen (N2) ice mixtures to energetic electrons. Prismatic P3N3 was detected in the gas phase and discriminated from its isomers utilizing isomer selective, tunable soft photoionization reflectron time-of-flight mass spectrometry during sublimation of the ices along with an isomer-selective photochemical processing converting prismatic P3N3 to 1,2,4-triaza-3,5,6-triphosphabicyclo[2.2.0]hexa-2,5-diene (P3N3). In prismatic P3N3, the P–P, P–N, and N–N bonds are lengthened compared to those in, e.g., diphosphine (P2H4), di-anthracene stabilized phosphorus mononitride (PN), and hydrazine (N2H4), by typically 0.03–0.10 Å. These findings advance our fundamental understanding of the chemical bonding of poly-nitrogen and poly-phosphorus systems and reveal a versatile pathway to produce exotic, ring-strained cage molecules.
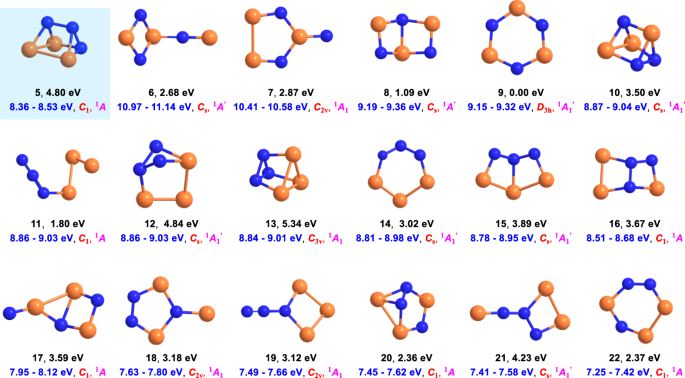
Spectroscopic Characterization and Photochemistry of the Vinylsulfinyl Radical.
Zhuang Wu, Lina Wang, Bo Lu, André K. Eckhardt, Peter R. Schreiner and Xiaoqing Zeng
PCCP 2021, 23,16307–16315. DOI: 10.1039/d1cp02584h.
The simplest α,β-unsaturated sulfinyl radical CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(H)SO˙ has been generated in the gas phase by high-vacuum flash pyrolysis (HVFP) of sulfoxide CH2
C(H)SO˙ has been generated in the gas phase by high-vacuum flash pyrolysis (HVFP) of sulfoxide CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(H)S(O)CF3 at ca. 800 °C. Two planar cis and trans conformers of CH2
C(H)S(O)CF3 at ca. 800 °C. Two planar cis and trans conformers of CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(H)SO˙ were isolated in cryogenic matrixes (N2, Ne, and Ar) and characterized with IR and UV/Vis spectroscopy. In addition to the photo-induced cis
C(H)SO˙ were isolated in cryogenic matrixes (N2, Ne, and Ar) and characterized with IR and UV/Vis spectroscopy. In addition to the photo-induced cis ![[leftrightharpoons]](https://www.rsc.org/images/entities/char_21cb.gif) trans conformational interconversion, CH2
trans conformational interconversion, CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(H)SO˙ displays complex photochemistry. Upon irradiation with a purple light LED (400 nm), CH2
C(H)SO˙ displays complex photochemistry. Upon irradiation with a purple light LED (400 nm), CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(H)SO˙ isomerizes to novel radicals CH3SCO˙, ˙CH2SC(O)H, and ˙CH2C(O)SH with concomitant dissociation to a caged molecular complex CH3S˙⋯CO. Subsequent UV-laser (266 nm) irradiation causes fragmentation to ˙CH3/OCS and additional formation of an elusive carbonyl radical CH3C(O)S˙, which rearranges to ˙CH2C(O)SH upon further UV-light irradiation (365 nm). The vibrational data and bonding analysis of the two conformers of CH2
C(H)SO˙ isomerizes to novel radicals CH3SCO˙, ˙CH2SC(O)H, and ˙CH2C(O)SH with concomitant dissociation to a caged molecular complex CH3S˙⋯CO. Subsequent UV-laser (266 nm) irradiation causes fragmentation to ˙CH3/OCS and additional formation of an elusive carbonyl radical CH3C(O)S˙, which rearranges to ˙CH2C(O)SH upon further UV-light irradiation (365 nm). The vibrational data and bonding analysis of the two conformers of CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(H)SO˙ suggest that both are floppy radicals in which the unpaired electron conjugates with the vicinal π(C
C(H)SO˙ suggest that both are floppy radicals in which the unpaired electron conjugates with the vicinal π(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C) bond, leading to significant contribution of the canonical resonance form of ˙CH2–C(H)SO. The mechanism for the isomerization of CH2
C) bond, leading to significant contribution of the canonical resonance form of ˙CH2–C(H)SO. The mechanism for the isomerization of CH2![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(H)SO˙ is discussed based on the observed intermediates along with a computed potential energy profile at the CCSD(T)-F12a/aug-cc-pVTZ//B3LYP/6-311++G(3df,3pd) level of theory.
C(H)SO˙ is discussed based on the observed intermediates along with a computed potential energy profile at the CCSD(T)-F12a/aug-cc-pVTZ//B3LYP/6-311++G(3df,3pd) level of theory.
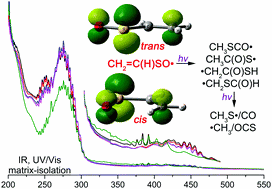
1,1,2-Ethenetriol: The Enol of Glycolic Acid, a High-Energy Prebiotic Molecule.
Artur Mardyukov, Felix Keul, and Peter R. Schreiner
Angew. Chem. Int. Ed. 2021, 60,15313–15316. DOI: 10.1002/anie.202104436
The simple yet uncharacterized high energy tautomer of glycolic acid, namely 1,1,2-ethenetriol, an important molecule in the abiotic synthesis of sugar acids, has now been identified by IR and UV/Vis spectroscopy. With these data now in hand, it now awaits its identification in space as another building block for prebiotic chemistry.
As low-temperature conditions (e.g. in space) prohibit reactions requiring large activation energies, an alternative mechanism for follow-up transformations of highly stable molecules involves the reactions of higher energy isomers that were generated in a different environment. Hence, one working model for the formation of larger organic molecules is their generation from high-lying isomers of otherwise rather stable molecules. As an example, we present here the synthesis as well as IR and UV/Vis spectroscopic identification of the previously elusive 1,1,2-ethenetriol, the higher energy enol tautomer of glycolic acid, a rather stable and hence unreactive biological building block. The title compound was generated in the gas phase by flash vacuum pyrolysis of tartronic acid at 400 °C and was subsequently trapped in argon matrices at 10 K. The spectral assignments are supported by B3LYP/6–311++G(2d,2p) computations. Upon photolysis at λ=180–254 nm, 1,1,2-ethenetriol rearranges to glycolic acid and ketene.
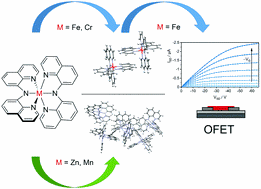
New π-Stacking Motifs for Molecular Semiconducting Materials: Bis(bis(8-quinolinyl)amide)metal(II) Complexes of Cr, Mn, Fe, and Zn.
Georg Albrecht, Harald Locke, Jonathan Becker, Limei Chen, Pascal Schweitzer, Peter R. Schreiner and Derck Schlettwein
Mat. Adv. 2021, 2, 2347–2357. DOI: 10.1039/D1MA00078K.
π–π stacking of adjacent molecules is an essential prerequisite for charge carrier transport in organic semiconductors. Neutral metal–organic complexes with two pincer-type bis(8-quinolinyl)amide (BQA) ligands forming orthogonal π-systems in complexes with octahedrally coordinated metal centres (Cr, Mn, Fe, Zn) were synthesized. Cr and Fe are shown to facilitate face-to-edge and parallel displaced stacking in two orthogonal directions as evident from single crystal X-ray diffraction (XRD). We demonstrate that the crystal structure as well as properties of the electron system of these complexes can be changed substantially upon variation of the metal centre. Cyclic voltammetry, UV-Vis absorption, and DFT computations were employed to characterize electronic properties at the molecular level. Thin films of the complexes, grown as interconnected islands, were prepared and investigated by optical spectroscopy, atomic force microscopy, and electrical measurements in organic field-effect transistor geometry. Increased conductivity was measured for thin films of the Fe and Cr complexes, which showed the strongest intermolecular coupling by optimized stacking in the independently grown single crystals. Successful transfer of such beneficial stacking into the thin films is discussed based on a combination of XRD and Raman spectroscopy.
Formation of Phosphine Imide (HN=PH3) and its Phosphinous Amide (H2N–PH2) Isomer.
Cheng Zhu, Alexandre Bergantini, Santosh K. Singh, Ralf I. Kaiser, André K. Eckhardt and Peter R. Schreiner
Chem. Commun. 2021, 57, 4958–4961. DOI: 10.1039/D0CC08411E
We present the first formation of the previously elusive phosphine imide (HN![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) PH3) along with its phosphinous amide (H2N–PH2) isomer via exposure of phosphine (PH3) and ammonia (NH3) ices to ionizing radiation. Our approach may be extended to prepare, separate, and detect highly reactive compounds such as intermediates of Wittig reactions.
PH3) along with its phosphinous amide (H2N–PH2) isomer via exposure of phosphine (PH3) and ammonia (NH3) ices to ionizing radiation. Our approach may be extended to prepare, separate, and detect highly reactive compounds such as intermediates of Wittig reactions.
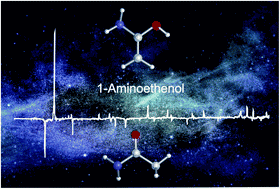
Preparation and Characterization of the Enol of Acetamide: 1-Aminoethenol, a High-Energy Prebiotic Molecule
Artur Mardyukov, Felix Keul, and Peter R. Schreiner
Chem. Sci. 2020, 11, 12358-12363. DOI: 10.1039/D0SC04906A
Amide tautomers, which constitute the higher-energy amide bond linkage, not only are key for a variety of biological but also prebiotic processes. In this work, we present the gas-phase synthesis of 1-aminoethenol, the higher-energy tautomer of acetamide, that has not been spectroscopically identified to date. The title compound was prepared by flash vacuum pyrolysis of malonamic acid and was characterized employing matrix isolation infrared as well as ultraviolet/visible spectroscopy. Coupled-cluster computations at the AE-CCSD(T)/cc-pVTZ level of theory support the spectroscopic assignments. Upon photolysis at λ > 270 nm, the enol rearranges to acetamide as well as ketene and ammonia. As the latter two are even higher in energy, they constitute viable starting materials for formation of the title compound.
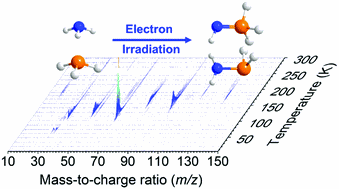
Interstellar Formation of Biorelevant Pyruvic Acid (CH3COCOOH)
Nils Fabian Kleimeier, André K. Eckhardt, Peter R. Schreiner and Ralf I. Kaiser
Chem 2020, 6-12, 3385-3395. DOI: 10.1016/j.chempr.2020.10.003
One of the key questions is how life could have emerged on early Earth and what chemicals and key reactions were involved. Terrestrial biomolecules, such as DNA, RNA, and peptides, formed from building blocks like nucleobases and amino acids. But where do these come from? Simple chemical building blocks could have formed on icy grains in space and may have survived comet impact on the early Earth. Pyruvic acid is widely accepted as a key prebiotic starting material, as it may have served as a fundamental building block for biorelevant molecules. This is underlined by the identification of pyruvic acid in carbonaceous meteorites. This study investigates the formation of pyruvic acid under interstellar conditions to encourage scientists in other fields to consider pyruvic acid as a potential interstellar molecule and include it in their radio-astronomical line searches. For chemists, the study will lead to a better understanding of the fundamental processes of abiotic syntheses of organic molecules.
Spectroscopic Identification of the •SSNO Isomers
Lina Wang, Zhuang Wu, Bo Lu, André K. Eckhardt, Peter R. Schreiner, Tarek Trabelsi, Joseph S. Francisco, Qian Yao, Hua Guo and Xiaoqing Zeng
J. Chem. Phys. 2020, 153, 094303. DOI: 10.1063/5.0020669
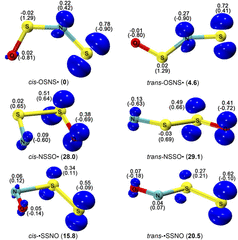 Elusive [S, S, N, O] isomers including the perthiyl radical •SSNO are S/N hybrid species in the complex bioinorganic chemistry of signaling molecules H2S and •NO. By mixing thermally generated disulfur (S2) with •NO in the gas phase, •SSNO was generated and subsequently isolated in cryogenic Ar- and N2-matrices at 10.0 K and 15.0 K, respectively. Upon irradiation with a 266 nm laser, •SSNO isomerizes to novel sulfinyl radicals cis-NSSO• and trans-NSSO• as well as thiyl radicals cis-OSNS• and trans-OSNS•, which have been characterized by combining matrix-isolation IR (15N-labeling) and UV/Vis spectroscopy and quantum chemical calculations at the CCSD(T)-F12/cc-pVTZ-F12 level of theory. The photo-induced reversible interconversion between NSSO• and OSNS• has also been observed.
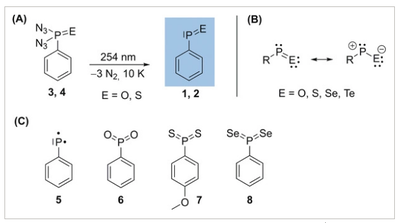
Elusive [S, S, N, O] isomers including the perthiyl radical •SSNO are S/N hybrid species in the complex bioinorganic chemistry of signaling molecules H2S and •NO. By mixing thermally generated disulfur (S2) with •NO in the gas phase, •SSNO was generated and subsequently isolated in cryogenic Ar- and N2-matrices at 10.0 K and 15.0 K, respectively. Upon irradiation with a 266 nm laser, •SSNO isomerizes to novel sulfinyl radicals cis-NSSO• and trans-NSSO• as well as thiyl radicals cis-OSNS• and trans-OSNS•, which have been characterized by combining matrix-isolation IR (15N-labeling) and UV/Vis spectroscopy and quantum chemical calculations at the CCSD(T)-F12/cc-pVTZ-F12 level of theory. The photo-induced reversible interconversion between NSSO• and OSNS• has also been observed.Isolation and Characterization of the Free Phenylphosphinidene Chalcogenides C6H5P=O and C6H5P=S, the Phosphorous Analogues of Nitrosobenzene and Thionitrosobenzene
Artur Mardyukov, Felix Keul and Peter R. Schreiner
Angew. Chem. Int. Ed. 2020, 59, 12445–12449. DOI: 10.1002/anie.202004172
The structures and reactivities of organic phosphinidene chalcogenides have been mainly inferred from trapping or complexation experiments. Phosphinidene chalcogenide derivatives appear to be an elusive family of molecules that have been suggested as reactive intermediates in multiple organophosphorus reactions. The quest to isolate “free” phosphinidene chalcogenides remains a challenge in the field. Here, we present the synthesis, IR, and UV/Vis spectroscopic identification of hitherto elusive phenylphosphinidene oxide and phenylphosphinidene sulfide from the corresponding phosphonic diazide precursors. We isolated these higher congeners of nitroso‐ and thionitrosobenzene in argon matrices at 10 K. The spectral assignments are supported by B3LYP/6–311++G(3df,3pd) and MP2/cc‐pVTZ computations.
Photochemistry of HNSO2 in Cryogenic Matrices: Spectroscopic Identification of the Intermediates and Mechanism
Changyun Chen, Lina Wang, Xiaofang Zhao, Zhuang Wu, Bastian Bernhardt, André K. Eckhardt, Peter R. Schreiner, Xiaoqing Zeng
PCCP 2020, 22, 7975–7983. DOI: 10.1039/D0CP00962H
Small molecules solely consisting of H, N, O, and S are highly relevant intermediates in atmospheric chemistry and biology. Even though several isomers of [HNO2S] have been computationally predicted, only the IR spectra for the two lowest-energy isomers HNSO2 and syn–syn HONSO have been previously reported. Herein, the photochemistry (193 nm laser) of HNSO2 in N2-, Ne-, and Ar-matrices (≤15 K) has been studied. Aside from syn–syn HONSO, several new isomers including anti–syn HONSO, gauche–syn HOSNO, syn HOS(O)N, anti HOS(O)N, syn HS(O)NO, anti HN(O)SO, gauche–syn HSONO, and an elusive caged-radical pair HOS˙⋯˙NO have been identified. Additionally, the formation of fragments HONO, HO˙, ˙NO, and ˙NO2 has also been observed. The characterization of these species with matrix-isolation IR and UV/Vis spectroscopy is supported by 15N-labeling and quantum chemical computations at the B3LYP/6-311++G(3df,3pd) level. Furthermore, the photo-induced isomerization reactions, including the conformational conversion of syn–syn HONSO → anti–syn HONSO and reversible isomerization of HOSNO ↔ anti–syn HONSO, syn–syn HONSO ↔ HN(O)SO, HSONO ↔ HS(O)NO, and HOS˙⋯˙NO ↔ HOSNO have also been observed, and the underlying mechanism is discussed.
The Elusive Cyclotriphosphazene Molecule and its Dewar-Benzene Type Valence Isomer (P3N3)
Cheng Zhu, André K. Eckhardt, Alexandre Bergantini, Santosh K. Singh, Peter R. Schreiner, Ralf I. Kaiser
Sci. Adv. 2020, 6, eaba6934. DOI: 10.1126/sciadv.aba6934
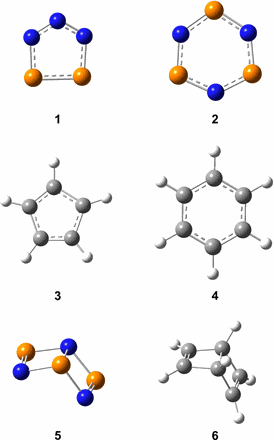
Although the chemistry of phosphorus and nitrogen has fascinated chemists for more than 350 years, the Hückel aromatic cyclotriphosphazene (P3N3, 2) molecule—a key molecular building block in phosphorus chemistry—has remained elusive. Here, we report a facile, versatile pathway producing cyclotriphosphazene and its Dewar benzene–type isomer (P3N3, 5) in ammonia-phosphine ices at 5 K exposed to ionizing radiation. Both isomers were detected in the gas phase upon sublimation via photoionization reflectron time-of-flight mass spectrometry and discriminated via isomer-selective photochemistry. Our findings provide a fundamental framework to explore the preparation of inorganic, isovalent species of benzene (C6H6) by formally replacing the C─H moieties alternatingly through phosphorus and nitrogen atoms, thus advancing our perception of the chemical bonding of phosphorus systems.
Capture and reactivity of an elusive carbon-sulfur centered biradical
Dennis Gerbig, Bastian Bernhardt, Raffael C. Wende and Peter R. Schreiner
J. Phys. Chem. 2020, 124, 2014–2018. DOI: 10.1021/acs.jpca.9b11795
The initial oxidation product of dimethyl sulfide in the marine boundary layer, the methyl thiomethyl radical, has remained elusive. A structurally analogous biradical with one radical center in the α-position to a sulfur atom could now be obtained by UV irradiation of p-nitrobenzaldehyde dithiane isolated in solid dinitrogen (N2) or Ar at cryogenic temperatures. A spin-forbidden reaction with triplet dioxygen (3O2) does not occur. The dithiane of o-nitrobenzaldehyde rather undergoes a series of rearrangements under the same conditions, resulting in overall photodeprotection.
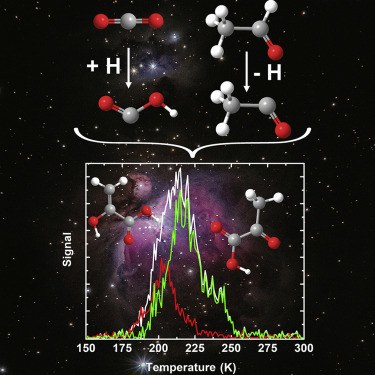
1,1-Ethenediol – The Long Elusive Enol of Acetic Acid
Artur Mardyukov, André K. Eckhardt, and Peter R. Schreiner
Angew. Chem. Int. Ed. 2020, 58, 5577–5580. DOI: 10.1002/anie.201915646
We present the first spectroscopic identification of hitherto unknown 1,1‐ethenediol, the enol tautomer of acetic acid. The title compound was generated in the gas phase through flash vacuum pyrolysis of malonic acid at 400 °C. The pyrolysis products were subsequently trapped in argon matrices at 10 K and characterized spectroscopically by means of IR and UV/Vis spectroscopy together with matching its spectral data with computations at the CCSD(T)/cc‐pCVTZ and B3LYP/6–311++G(2d,2p) levels of theory. Upon photolysis at λ=254 nm, the enol rearranges to acetic acid and ketene.
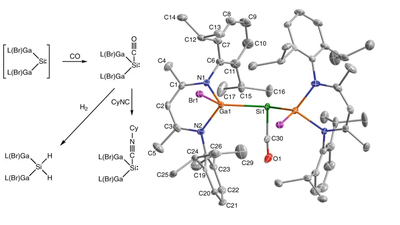
A silicon–carbonyl complex stable at room temperature
Chelladurai Ganesamoorthy, Juliane Schoening, Christoph Wölper, Lijuan Song, Peter R. Schreiner, and Stephan Schulz
Nat. Chem. 2020, 12, 608–614. DOI: 10.1038/s41557-020-0456-x
Highlights: a) Leigh Krietsch Boerner Chem. Eng. News. 2020, 98 (16), 9; b) David Schilter Nat. Rev. Chem. 2020, 4, 274; c) Nachr. Chem. 2020, 68, 42.
Main-group-element compounds with energetically high-lying donor and low-lying acceptor orbitals are able to mimic chemical bonding motifs and reactivity patterns known in transition metal chemistry, including small-molecule activation and catalytic reactions. Monovalent group 13 compounds and divalent group 14 compounds, particularly silylenes, have been shown to be excellent candidates for this purpose. However, one of the most common reactions of transition metal complexes, the direct reaction with carbon monoxide and formation of room-temperature isolable carbonyl complexes, is virtually unknown in main-group-element chemistry. Here, we show the synthesis, single-crystal X-ray structure, and density functional theory computations of a room-temperature-stable silylene carbonyl complex [L(Br)Ga]2Si:–CO (L = HC[C(Me)N(2,6-iPr2-C6H3)]2), which was obtained by direct carbonylation of the electron-rich silylene intermediate [L(Br)Ga]2Si:. Furthermore, [L(Br)Ga]2Si:–CO reacts with H2 and PBr3 with bond activation, whereas the reaction with cyclohexyl isocyanide proceeds with CO substitution.
Spectroscopic Identification of the Phenyltelluryl Radical and its Reactivity Toward Molecular Oxygen
Felix Keul, Artur Mardyukov and Peter R. Schreiner
PCCP 2019, 21, 25797–25801. DOI: 10.1039/C9CP05112K
The phenyltelluryl radical was prepared by high-vacuum flash pyrolysis of diphenyl ditelluride and was chacracterized by matrix isolation IR and UV/Vis spectroscopy. After doping the matrix with molecular oxygen and allowing bimolecular reactions, the hitherto unkown phenyltelluro peroxy radical formed and was identified via IR spectroscopy. Irradiation with light at λ = 436 nm leads to isomerization to the thermodynamically more stable novel phenyltelluroyl radical. All experimental findings agree well with density functional theory (UB3LYP/Def2QZVPP and UM06-2X/Def2QZVPP) computations.